Contemporary management of renal angiomyolipomas
Renal angiomyolipomas (AMLs) are benign neoplasms composed of adipose tissue, smooth muscle and blood vessels classified under neoplasms of the perivascular epithelioid cells (PEComas) (1). AMLs can typically be diagnosed by imaging alone due to its characteristic attenuation of tumor containing fat on Computer Tomography scan (CT). AMLs are either sporadic or associated with tuberous sclerosis complex (TSC) or lymphangioleiomyomatosis (LAM). TSC is a multisystem autosomal dominant disorder classically described as the triad of epilepsy, mental retardation and adenoma sebaceum (2,3). It is transmitted as an autosomal dominant trait in 20% to 30% of patients with the remaining patients developing this complex as a sporadic mutation (4,5). Mutations in the tumor suppressor genes TSC1 (9q34), which encodes for the protein hartin or TSC2 (16p13), which encodes for the protein tuberin result in TSC (6-8). These genes function as a complex to inhibit the mechanistic target of rapamycin (mTOR) pathway, which is important in the regulation of cell growth, migration, and proliferation. In patients with TSC, renal manifestations include AML (40% to 80%), renal cysts (20%) and Renal Cell Carcinoma (RCC) (2%). AMLs in TSC are often multiple, bilateral and are faster growing compared to sporadic cases (9-12). LAM is a rare, progressive, multisystem disorder primarily resulting in cystic lung disease and has a predilection for women (13-16). It is associated with TSC about 30% of the time, known as TSC-LAM. The remaining cases are known as sporadic LAM (S-LAM) (17-19). Sporadic cases of AML account for about 80% of cases with a prevalence of 0.01% to 0.3% (20-22) and typically occur in middle-aged women (12). One hypothesis that potentially explains the predisposition of sporadic AMLs in females, AML growth in patients on hormonal therapy (23) and during pregnancy (24,25) is the ubiquitous expression of estrogen receptor beta, progesterone receptor and androgen receptor (26-28).
AMLs are the most common renal tumor associated with spontaneous hemorrhage in young patients with up to 20% of patients presenting with shock (29). AMLs can also cause a wide spectrum of symptoms including hematuria, pain or impairment of renal function. Treatment of AMLs includes surgery (nephrectomy or partial nephrectomy), selective angioembolization, thermal ablation, or systemic therapy with mTOR (mammalian target of rapamycin) inhibitors (11,30-40). The principal rationale for treatment of asymptomatic AML is to prevent life threatening bleeding or malignant transformation, both of which are rare events.
Clinicians must carefully balance the risks of treatment versus observation for individual patients. For young patients with large tumors or for patients who cannot be followed, surgery remains an excellent option, especially when a minimally invasive approach can be used. The principal benefit of surgery compared to other modalities is removal of all AML tissue, lowering the risk of future hemorrhage. However, surgery has an associated 0.3% to 1.7% risk of major complication (33,41) and patients may have longer recovery compared to other treatments.
Selective angioembolization is a minimally invasive option for AML treatment, which preserves renal function (42) and has low rates of procedural complications. However, AMLs may not shrink significantly after angioembolization, and it’s unclear if the risk of hemorrhage remains for tumors that remain >4 cm and therefore retreatment is not uncommon. Kothary et al., reported a 32% recurrence rate in AML lesions >4 cm, only recurring in patients with TSC (no patients with sporadic AML) with a median follow up of 51 months (36). Similarly, Bishay et al., performed angioembolization on AMLs >10 cm with 38% requiring multiple sessions with a follow up of 29 months (43).
Thermal ablation, which includes radiofrequency ablation (RFA), microwave ablation, and cryoablation are minimally invasive modalities that can be used to treat AMLs laparoscopically or percutaneously. Several series have demonstrated that ablation is a safe option for treatment of AMLs (32,44-46), however, there is a lack of data demonstrating long-term treatment success with this modality. Johnson et al., reported 3 patients with AMLs in solitary kidneys who safely underwent CT guided percutaneous cryoablation. The AMLs were 1.2–2.5 cm with no procedural or postoperative complications with a follow up of 5–36 months and no evidence of radiographic recurrence in two of the patients, however, the third patient did display persistent AML (47). In a study of AMLs treated with RFA with a mean size of 9.5 cm (range, 9–19 cm), Sooriakumaran et al., reported retreatment of three of the five AMLs. When compared to RFA, microwave ablation has theoretical advantages of larger ablative zones from a decreased heat sink effect and from synergy among multiple probes (48,49). In a series by Cristescu et al., seven patients were treated with microwave ablation for 11 renal AMLs with a mean size of 3.4 cm (range, 2.4–4.9 cm). All the ablations were technically successful with no major complications. There was a decrease in diameter and volume of 29% and 47% respectively at a median follow up of 23.1 months (50).
Systemic therapy using mTOR inhibitors is another treatment used for AMLs in patients with TSC and/or LAM (51). TSC results in mutations in the hamartin (TSC1) and tuberin (TSC2) gene, which form a complex that regulates the activity of MTOR1. MTOR1 regulates cell proliferation and growth and when this is no longer regulated as observed in patients with mutations in TSC1 or TSC2, this results in hamartomas in the brain, lung, kidney skin and heart (52). MTOR inhibitors, such as Sirolimus, work as an immunosuppressive agent, which forms a complex with FK binding protein 12 inactivating the target of MTOR1 and inhibiting downstream signaling (39,40,53,54). A systematic review of four prospective nonrandomized studies involving 94 patients demonstrated an overall response rate to Sirolimus of 46.8% (44 of 94 patients) in the first year. In the second year for those patients still being treated, the response rate was 43.5% (20 of 46 patients). The most common side effects were stomatitis, respiratory infection, skin lesions and hyperlipidemia with rare serious adverse side effects. The authors concluded that AMLs did shrink during Sirolimus therapy and treatment was safe, however, they did tend to regrow after therapy was stopped (51). A second systematic review of randomized or quasi-randomized studies of Sirolimus or Everolimus (FDA approved) involving 263 patients demonstrated that treatment resulted in a 50% reduction in size of AMLs (55).
While asymptomatic AMLs smaller than 4 cm are generally observed with serial imaging, active treatment has generally been recommended for symptomatic AMLs and AMLs greater than 4 cm in diameter when patients have: (I) risk of hemorrhage (especially in pregnant women or women of child-bearing age); (II) increased risk of aneurysm formation; (III) poor access to follow up imaging (56-58). These recommendations stem from several studies reporting that AMLs larger than 4 cm are more likely to be symptomatic compared to AMLs less than 4 cm (59-65). Oesterling et al. performed a literature review incorporating data from 1948 to 1985 reviewing 602 AMLs and determined that of patients that had AMLs larger than 4 cm in diameter, 86% were symptomatic with 9% of patients presenting in hemorrhagic shock at time of presentation. However, patients with tumors less than 4 cm in diameter were only symptomatic 23% of the time (66). Additionally, it was observed that AMLs less than 4 cm did not grow at all or grew at a slower rate compared to AMLs that were larger than 4 cm (63,66).
More recently, a large study has suggested that some asymptomatic AMLs >4 cm do not need to be treated and could be followed safely with surveillance. Bhatt et al. retrospectively analyzed 582 AMLs in 447 patients with a median follow up of 43 months and determined that 32% of AMLs >4 cm were symptomatic compared to 6.6% of AMLs ≤4 cm. Notably, only 2 cases (0.4%) required emergency intervention due to bleeding. In both of these cases, the patients were female with aneurysms >13 cm who were subsequently managed by arterial embolization. Unlike prior studies, there was no difference in growth rates for AMLs >4 cm in diameter when compared to smaller AMLs. Most importantly, the vast majority (91%) of AMLs had no growth or grew very slowly with an average growth rate of 0.02 cm/year. In 25 AMLs that were actively treated, patients were more likely to be young, and have symptomatic tumors with an initial size of >4 cm. However, most patients (70%) with AMLs >4 cm were asymptomatic and 61.7% of AMLs >4 cm were managed conservatively with serial imaging. Of the AMLs >4 cm that were intervened upon, 50% were asymptomatic and 16 of the 18 cases were performed electively. The authors therefore concluded that a strict 4 cm size cutoff should not be an indication for active treatment.
It is important to note that sporadic AMLs and TSC associated AMLs behave very differently and therefore should be approached differently. AMLs associated with TSC compared to sporadic cases tend to grow at a faster rate (1.25 vs. 0.19 cm respectively) (12) and have a higher risk of malignant transformation (67). In the study by Bhatt et al., 2 of the 25 cases that were intervened upon were for metastatic epithelioid AMLs. These epithelioid variants also behave differently than typical AMLs. These were treated with mammalian target of rapamycin (MTOR inhibitors). The 2004 World Health Organization (WHO) Classification characterizes epithelioid-AML by a proliferation of predominantly epithelioid cells with malignant potential (68). It is a rare subtype of AML, which is exemplified by one series consisting of 437 AMLs with only 4.6% classified as epithelioid-AML. In this same series, which had a mean follow up of 82.5 months, only 1 patient (5%) developed distant metastases (69). A second case series by Aydin et al. reviewed 194 AMLs resected at the Cleveland Clinic between 1981 and 2007 reported an incidence of epithelioid-AML of 7.7%. With a mean follow up of 5.1 years, no patients had evidence of metastatic disease (67). In terms risk factors for local recurrence or metastatic behavior, two case series consisting of 41 cases and 20 cases predicted size greater than 7.7 cm, >20% epithelioid histology, enlarged vesicular nuclei with prominent nucleoli, size of 19 cm, presence of TSC, tumor necrosis, extrarenal extension or renal vein invasion and carcinoma-like histology respectively as risk factors for local recurrence or metastatic behavior (70,71).
Renal AMLs are rare tumors and there are no prospective randomized controlled trials available to evaluate active treatment or surveillance. Ouzaid et al. showed that 13 of 38 patients failed active surveillance of AMLs with size >4 cm and demonstrated that symptoms at presentation was associated with failure of active surveillance during a mean follow up of 40 months. However, if all the tumors in the study were treated by size >4 cm criteria alone, 67% of the patients would have undergone “unnecessary” treatment (72). In a pooled analysis reviewing 441 patients from 58 studies and 3 institutions, the risk of bleeding increased from 10% to 24% in tumors that were on average 4 and 6 cm respectively, suggesting that a 4 cm cutoff may be too conservative. Therefore, active surveillance of AMLs is an excellent option for patients with AML <4 cm. Interestingly, the recent study by Bhatt et al. suggests that this strategy could also be used for patients with larger AMLs (10).
Clinicians who evaluate patients with AMLs are likely familiar with active surveillance for patients with low risk prostate cancer (73) or small RCCs (74) and can be managed similarly to these other urologic conditions. In low risk cancers, active surveillance has been used as a strategy to limit overall morbidity by avoiding treatment for most patients. Low risk prostate cancer and RCC are ideal for surveillance strategies since these cancers primarily occur in older patients who may have other competing causes of mortality. In addition, most low risk tumors progress slowly over time, which allows for delayed treatment in a subset of patients who progress either by increase in tumor size (75) or tumor markers (76). Interestingly, surveillance of larger AMLs may also have unique challenges when compared to low risk RCC or prostate cancer. For example, most AMLs occur in younger women who have significant expectations for longevity [the median age of diagnosis was 52 years old for AML>4 cm in Bhatt et al. (10)]. In 2016, a 52-year-old woman in the United States can expect to live an average of 33.5 additional years (77). Extensive monitoring with serial imaging is costly and exposes the patient to risks of ionizing radiation from multiple CT scans (78). It is unclear how the risk of bleeding from AML changes over time and there are no reliable characteristics to predict increased risk of hemorrhage during observation. Historically, studies of AML have estimated bleeding risk from on single linear measurements of asymmetric three-dimensional lesions instead of tumor volume, vascularity or other objective measurements. Bhatt et al. demonstrated that AML >4 cm grow at similar rates to smaller AML (10), further suggesting that growth rate is a poor predictor of the risk of future hemorrhage.
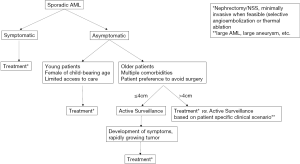
With the available studies on the natural history of AMLs, there are many uncertainties and it is not possible to form strong evidence based treatment guidelines. Recent data has suggested that it is not necessary to actively treat all AMLs >4 cm. The overall risk of hemorrhage appears to be low and most patients were managed safely with initial active surveillance in a large retrospective study (10). However, without reliable methods to predict risk, long-term surveillance may be problematic in young patients who are otherwise healthy. Active treatment of larger asymptomatic AMLs should continue for women of childbearing age or those patients without reliable access to medical care. With these considerations, we have composed a general guideline for treatment of AMLs shown in Figure 1. Future prospective studies should evaluate protocols for surveillance and determine the most efficient approach for treatment of patients with AML >4 cm.
Acknowledgments
Funding: None.
Footnote
Provenance and Peer Review: This article was commissioned and reviewed by the Section Editor Hong-Chao He, MD, PhD (Department of Urology, Shanghai Ruijin Hospital, Shanghai Jiaotong University School of Medicine, Shanghai, China).
Conflicts of Interest: Both authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/tcr.2016.11.57). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Martignoni G, Pea M, Reghellin D, et al. PEComas: the past, the present and the future. Virchows Arch 2008;452:119-32. [Crossref] [PubMed]
- Lagos JC, Gomez MR. Tuberous sclerosis: reappraisal of a clinical entity. Mayo Clin Proc 1967;42:26-49. [PubMed]
- Pampiglione G, Moynahan EJ. The tuberous sclerosis syndrome: clinical and EEG studies in 100 children. J Neurol Neurosurg Psychiatry 1976;39:666-73. [Crossref] [PubMed]
- Eble JN. Angiomyolipoma of kidney. Semin Diagn Pathol 1998;15:21-40. [PubMed]
- Neumann HP, Schwarzkopf G, Henske EP. Renal angiomyolipomas, cysts, and cancer in tuberous sclerosis complex. Semin Pediatr Neurol 1998;5:269-75. [Crossref] [PubMed]
- Jones AC, Shyamsundar MM, Thomas MW, et al. Comprehensive mutation analysis of TSC1 and TSC2-and phenotypic correlations in 150 families with tuberous sclerosis. Am J Hum Genet 1999;64:1305-15. [Crossref] [PubMed]
- Dabora SL, Jozwiak S, Franz DN, et al. Mutational analysis in a cohort of 224 tuberous sclerosis patients indicates increased severity of TSC2, compared with TSC1, disease in multiple organs. Am J Hum Genet 2001;68:64-80. [Crossref] [PubMed]
- Sancak O, Nellist M, Goedbloed M, et al. Mutational analysis of the TSC1 and TSC2 genes in a diagnostic setting: genotype--phenotype correlations and comparison of diagnostic DNA techniques in Tuberous Sclerosis Complex. Eur J Hum Genet 2005;13:731-41. [Crossref] [PubMed]
- Rakowski SK, Winterkorn EB, Paul E, et al. Renal manifestations of tuberous sclerosis complex: Incidence, prognosis, and predictive factors. Kidney Int 2006;70:1777-82. [Crossref] [PubMed]
- Bhatt JR, Richard PO, Kim NS, et al. Natural History of Renal Angiomyolipoma (AML): Most Patients with Large AMLs >4cm Can Be Offered Active Surveillance as an Initial Management Strategy. Eur Urol 2016;70:85-90. [Crossref] [PubMed]
- Nelson CP, Sanda MG. Contemporary diagnosis and management of renal angiomyolipoma. J Urol 2002;168:1315-25. [Crossref] [PubMed]
- Seyam RM, Bissada NK, Kattan SA, et al. Changing trends in presentation, diagnosis and management of renal angiomyolipoma: comparison of sporadic and tuberous sclerosis complex-associated forms. Urology 2008;72:1077-82. [Crossref] [PubMed]
- Kalassian KG, Doyle R, Kao P, et al. Lymphangioleiomyomatosis: new insights. Am J Respir Crit Care Med 1997;155:1183-6. [Crossref] [PubMed]
- Sullivan EJ. Lymphangioleiomyomatosis: a review. Chest 1998;114:1689-703. [Crossref] [PubMed]
- Workshop Summary NHLBI. Report of workshop on lymphangioleiomyomatosis. National Heart, Lung, and Blood Institute. Am J Respir Crit Care Med 1999;159:679-83. [Crossref] [PubMed]
- Johnson S. Rare diseases. 1. Lymphangioleiomyomatosis: clinical features, management and basic mechanisms. Thorax 1999;54:254-64. [Crossref] [PubMed]
- Costello LC, Hartman TE, Ryu JH. High frequency of pulmonary lymphangioleiomyomatosis in women with tuberous sclerosis complex. Mayo Clin Proc 2000;75:591-4. [Crossref] [PubMed]
- Franz DN, Brody A, Meyer C, et al. Mutational and radiographic analysis of pulmonary disease consistent with lymphangioleiomyomatosis and micronodular pneumocyte hyperplasia in women with tuberous sclerosis. Am J Respir Crit Care Med 2001;164:661-8. [Crossref] [PubMed]
- Moss J, Avila NA, Barnes PM, et al. Prevalence and clinical characteristics of lymphangioleiomyomatosis (LAM) in patients with tuberous sclerosis complex. Am J Respir Crit Care Med 2001;164:669-71. [Crossref] [PubMed]
- Hajdu SI, Foote FW Jr. Angiomyolipoma of the kidney: report of 27 cases and review of the literature. J Urol 1969;102:396-401. [PubMed]
- Fujii Y, Ajima J, Oka K, et al. Benign renal tumors detected among healthy adults by abdominal ultrasonography. Eur Urol 1995;27:124-7. [PubMed]
- Osborne JP, Fryer A, Webb D. Epidemiology of tuberous sclerosis. Ann N Y Acad Sci 1991;615:125-7. [Crossref] [PubMed]
- Gould Rothberg BE, Grooms MC, Dharnidharka VR. Rapid growth of a kidney angiomyolipoma after initiation of oral contraceptive therapy. Obstet Gynecol 2006;108:734-6. [Crossref] [PubMed]
- Fernández Arjona M, Mínguez R, Serrano P, et al. Rapidly-growing renal angiomyolipoma associated with pregnancy. Actas Urol Esp 1994;18:755-7. [PubMed]
- King JA, Stamilio DM. Maternal and fetal tuberous sclerosis complicating pregnancy: a case report and overview of the literature. Am J Perinatol 2005;22:103-8. [Crossref] [PubMed]
- Boorjian SA, Sheinin Y, Crispen PL, et al. Hormone receptor expression in renal angiomyolipoma: clinicopathologic correlation. Urology 2008;72:927-32. [Crossref] [PubMed]
- Henske EP, Ao X, Short MP, et al. Frequent progesterone receptor immunoreactivity in tuberous sclerosis-associated renal angiomyolipomas. Mod Pathol 1998;11:665-8. [PubMed]
- L'Hostis H, Deminiere C, Ferriere JM, et al. Renal angiomyolipoma: a clinicopathologic, immunohistochemical, and follow-up study of 46 cases. Am J Surg Pathol 1999;23:1011-20. [Crossref] [PubMed]
- Pode D, Meretik S, Shapiro A, et al. Diagnosis and management of renal angiomyolipoma. Urology 1985;25:461-7. [Crossref] [PubMed]
- Schwartz MJ, Smith EB, Trost DW, et al. Renal artery embolization: clinical indications and experience from over 100 cases. BJU Int 2007;99:881-6. [Crossref] [PubMed]
- Fazeli-Matin S, Novick AC. Nephron-sparing surgery for renal angiomyolipoma. Urology 1998;52:577-83. [Crossref] [PubMed]
- Prevoo W, van den Bosch MA, Horenblas S. Radiofrequency ablation for treatment of sporadic angiomyolipoma. Urology 2008;72:188-91. [Crossref] [PubMed]
- Boorjian SA, Frank I, Inman B, et al. The role of partial nephrectomy for the management of sporadic renal angiomyolipoma. Urology 2007;70:1064-8. [Crossref] [PubMed]
- Soulen MC, Faykus MH Jr, Shlansky-Goldberg RD, et al. Elective embolization for prevention of hemorrhage from renal angiomyolipomas. J Vasc Interv Radiol 1994;5:587-91. [Crossref] [PubMed]
- Han YM, Kim JK, Roh BS, et al. Renal angiomyolipoma: selective arterial embolization--effectiveness and changes in angiomyogenic components in long-term follow-up. Radiology 1997;204:65-70. [Crossref] [PubMed]
- Kothary N, Soulen MC, Clark TW, et al. Renal angiomyolipoma: long-term results after arterial embolization. J Vasc Interv Radiol 2005;16:45-50. [Crossref] [PubMed]
- Byrd GF, Lawatsch EJ, Mesrobian HG, et al. Laparoscopic cryoablation of renal angiomyolipoma. J Urol 2006;176:1512-6; discussion 1516. [Crossref] [PubMed]
- Delworth MG, Pisters LL, Fornage BD, et al. Cryotherapy for renal cell carcinoma and angiomyolipoma. J Urol 1996;155:252-4; discussion 254-5. [Crossref] [PubMed]
- Bissler JJ, Kingswood JC, Radzikowska E, et al. Everolimus for angiomyolipoma associated with tuberous sclerosis complex or sporadic lymphangioleiomyomatosis (EXIST-2): a multicentre, randomised, double-blind, placebo-controlled trial. Lancet 2013;381:817-24. [Crossref] [PubMed]
- Bissler JJ, Kingswood JC, Radzikowska E, et al. Everolimus for renal angiomyolipoma in patients with tuberous sclerosis complex or sporadic lymphangioleiomyomatosis: extension of a randomized controlled trial. Nephrol Dial Transplant 2016;31:111-9. [Crossref] [PubMed]
- Stephenson AJ, Hakimi AA, Snyder ME, et al. Complications of radical and partial nephrectomy in a large contemporary cohort. J Urol 2004;171:130-4. [Crossref] [PubMed]
- Ramon J, Rimon U, Garniek A, et al. Renal angiomyolipoma: long-term results following selective arterial embolization. Eur Urol 2009;55:1155-61. [Crossref] [PubMed]
- Bishay VL, Crino PB, Wein AJ, et al. Embolization of giant renal angiomyolipomas: technique and results. J Vasc Interv Radiol 2010;21:67-72. [Crossref] [PubMed]
- Jacomides L, Ogan K, Watumull L, et al. Laparoscopic application of radio frequency energy enables in situ renal tumor ablation and partial nephrectomy. J Urol 2003;169:49-53; discussion 53. [Crossref] [PubMed]
- Sooriakumaran P, Gibbs P, Coughlin G, et al. Angiomyolipomata: challenges, solutions, and future prospects based on over 100 cases treated. BJU Int 2010;105:101-6. [Crossref] [PubMed]
- Castle SM, Gorbatiy V, Ekwenna O, et al. Radiofrequency ablation (RFA) therapy for renal angiomyolipoma (AML): an alternative to angio-embolization and nephron-sparing surgery. BJU Int 2012;109:384-7. [Crossref] [PubMed]
- Johnson SC, Graham S, D'Agostino H, et al. Percutaneous renal cryoablation of angiomyolipomas in patients with solitary kidneys. Urology 2009;74:1246-9. [Crossref] [PubMed]
- Wen CC, Nakada SY. Energy ablative techniques for treatment of small renal tumors. Curr Opin Urol 2006;16:321-6. [Crossref] [PubMed]
- Laeseke PF, Lee FT Jr, Sampson LA, et al. Microwave ablation versus radiofrequency ablation in the kidney: high-power triaxial antennas create larger ablation zones than similarly sized internally cooled electrodes. J Vasc Interv Radiol 2009;20:1224-9. [Crossref] [PubMed]
- Cristescu M, Abel EJ, Wells S, et al. Percutaneous Microwave Ablation of Renal Angiomyolipomas. Cardiovasc Intervent Radiol 2016;39:433-40. [Crossref] [PubMed]
- Peng ZF, Yang L, Wang TT, et al. Efficacy and safety of sirolimus for renal angiomyolipoma in patients with tuberous sclerosis complex or sporadic lymphangioleiomyomatosis: a systematic review. J Urol 2014;192:1424-30. [Crossref] [PubMed]
- Roach ES, Sparagana SP. Diagnosis of tuberous sclerosis complex. J Child Neurol 2004;19:643-9. [PubMed]
- Kenerson HL, Aicher LD, True LD, et al. Activated mammalian target of rapamycin pathway in the pathogenesis of tuberous sclerosis complex renal tumors. Cancer Res 2002;62:5645-50. [PubMed]
- Kwiatkowski DJ, Zhang H, Bandura JL, et al. A mouse model of TSC1 reveals sex-dependent lethality from liver hemangiomas, and up-regulation of p70S6 kinase activity in Tsc1 null cells. Hum Mol Genet 2002;11:525-34. [Crossref] [PubMed]
- Sasongko TH, Ismail NF, Zabidi-Hussin Z. Rapamycin and rapalogs for tuberous sclerosis complex. Cochrane Database Syst Rev 2016;7:CD011272 [PubMed]
- Flum AS, Hamoui N, Said MA, et al. Update on the Diagnosis and Management of Renal Angiomyolipoma. J Urol 2016;195:834-46. [Crossref] [PubMed]
- Ljungberg B, Bensalah K, Canfield S, et al. EAU guidelines on renal cell carcinoma: 2014 update. Eur Urol 2015;67:913-24. [Crossref] [PubMed]
- Preece P, Mees B, Norris B, et al. Surgical management of haemorrhaging renal angiomyolipoma in pregnancy. Int J Surg Case Rep 2015;7C:89-92. [Crossref] [PubMed]
- Dickinson M, Ruckle H, Beaghler M, et al. Renal angiomyolipoma: optimal treatment based on size and symptoms. Clin Nephrol 1998;49:281-6. [PubMed]
- Kessler OJ, Gillon G, Neuman M, et al. Management of renal angiomyolipoma: analysis of 15 cases. Eur Urol 1998;33:572-5. [Crossref] [PubMed]
- Chen SS, Lin AT, Chen KK, et al. Renal angiomyolipoma--experience of 20 years in Taiwan. Eur Urol 1997;32:175-8. [PubMed]
- Koh KB, George J. Radiological parameters of bleeding renal angiomyolipoma. Scand J Urol Nephrol 1996;30:265-8. [Crossref] [PubMed]
- Kennelly MJ, Grossman HB, Cho KJ. Outcome analysis of 42 cases of renal angiomyolipoma. J Urol 1994;152:1988-91. [PubMed]
- van Baal JG, Smits NJ, Keeman JN, et al. The evolution of renal angiomyolipomas in patients with tuberous sclerosis. J Urol 1994;152:35-8. [PubMed]
- Steiner MS, Goldman SM, Fishman EK, et al. The natural history of renal angiomyolipoma. J Urol 1993;150:1782-6. [PubMed]
- Oesterling JE, Fishman EK, Goldman SM, et al. The management of renal angiomyolipoma. J Urol 1986;135:1121-4. [PubMed]
- Aydin H, Magi-Galluzzi C, Lane BR, et al. Renal angiomyolipoma: clinicopathologic study of 194 cases with emphasis on the epithelioid histology and tuberous sclerosis association. Am J Surg Pathol 2009;33:289-97. [Crossref] [PubMed]
- Humphrey PA, Moch H, Cubilla AL, et al. The 2016 WHO Classification of Tumours of the Urinary System and Male Genital Organs-Part B: Prostate and Bladder Tumours. Eur Urol 2016;70:106-19. [Crossref] [PubMed]
- He W, Cheville JC, Sadow PM, et al. Epithelioid angiomyolipoma of the kidney: pathological features and clinical outcome in a series of consecutively resected tumors. Mod Pathol 2013;26:1355-64. [Crossref] [PubMed]
- Nese N, Martignoni G, Fletcher CD, et al. Pure epithelioid PEComas (so-called epithelioid angiomyolipoma) of the kidney: A clinicopathologic study of 41 cases: detailed assessment of morphology and risk stratification. Am J Surg Pathol 2011;35:161-76. [Crossref] [PubMed]
- Brimo F, Robinson B, Guo C, et al. Renal epithelioid angiomyolipoma with atypia: a series of 40 cases with emphasis on clinicopathologic prognostic indicators of malignancy. Am J Surg Pathol 2010;34:715-22. [PubMed]
- Ouzaid I, Autorino R, Fatica R, et al. Active surveillance for renal angiomyolipoma: outcomes and factors predictive of delayed intervention. BJU Int 2014;114:412-7. [PubMed]
- Tosoian JJ, Carter HB, Lepor A, et al. Active surveillance for prostate cancer: current evidence and contemporary state of practice. Nat Rev Urol 2016;13:205-15. [Crossref] [PubMed]
- Pierorazio PM, Johnson MH, Patel HD, et al. Management of Renal Masses and Localized Renal Cancer: Systematic Review and Meta-Analysis. J Urol 2016;196:989-99. [Crossref] [PubMed]
- Jewett MA, Zuniga A. Renal tumor natural history: the rationale and role for active surveillance. Urol Clin North Am 2008;35:627-34. vii. [Crossref] [PubMed]
- Bruinsma SM, Bangma CH, Carroll PR, et al. Active surveillance for prostate cancer: a narrative review of clinical guidelines. Nat Rev Urol 2016;13:151-67. [Crossref] [PubMed]
- US Social Security Administration Life Expectancy Calculator]. Available online: https://www.ssa.gov/cgi-bin/longevity.cgi
- Fazel R, Krumholz HM, Wang Y, et al. Exposure to low-dose ionizing radiation from medical imaging procedures. N Engl J Med 2009;361:849-57. [Crossref] [PubMed]