The association between ephrin receptor-A1 expression and survival in patients with cancer: a meta-analysis
Introduction
Despite the advances in treatment modalities, the survival of patients with cancer is still poor. For this reason, individualized and targeted therapies have been developed, and research on biomarkers that can predict the prognosis of patients with cancer has been essential. Therefore, researchers have attempted to find molecules participating in cancer pathogenesis (1). Among them, Ephrin receptor was speculated to be a possible key factor in carcinogenesis and a biomarker of tumorigenic processes (1).
The Ephrin receptor was originally found in hepatocellular carcinoma cells in 1987 (2). Ephrin receptor-A1 (EPHA1) is a member of the Ephrin tyrosine kinase receptor family and is a plasma membrane protein (2). EPHA1 participates in various developmental processes by engaging in cell adhesion, migration, and tissue boundary formation. In addition to these biological functions, EPHA1 is involved in tumor angiogenesis, tumor invasion, and metastasis (2-4). Moreover, studies show that EPHA1 can not only predict the prognosis of patients with cancer but also serve as a target for cancer treatment (3,5-12).
However, the results of individual studies were inconsistent. Therefore, we evaluated the association between survival and EPHA1 expression in patients with cancer. We present the following article in accordance with the PRISMA reporting checklist (available at https://tcr.amegroups.com/article/view/10.21037/tcr-21-1367/rc).
Methods
Search strategy
Studies were selected through a literature search performed in PubMed, Embase, Scopus, and the Cochrane library until February 8, 2022, using the following keywords: (EPHA1 or Ephrin receptor-A1) and (cancer, tumor, carcinoma, neoplasm, or malignancy) and (prognostic or predict prognosis, survival, or outcome). A manual search was conducted through the references to related studies.
Inclusion and exclusion criteria
The studies were included in the analysis only if they met the following criteria: (I) EPHA1 expression was assessed in human cancer; (II) the association between survival and EPHA1 expression was evaluated by immunohistochemistry; (III) survival data were provided for the calculation of the hazard ratio (HR) with 95% confidence interval (CI). Studies were excluded if they met the following criteria: (I) duplicate-searched articles; (II) conference abstracts, reviews, and non-English articles.
Data extraction and quality assessment
Each of the two researchers individually reviewed the included studies and collected the following information: first author, publication year, country, cancer type, sample size, sex of patients, follow-up time, survival outcome, and cut-off value of EPHA1 expression. If there was any difference in the information collected, we reached consensus through discussion. Each of the two researchers evaluated the quality of the included studies using the Newcastle-Ottawa Scale. If there was any difference in the information collected, we reached an agreement regarding the quality of the study through discussion.
Statistical analysis
The pooled HR and odds ratio (OR) with 95% CI were calculated to evaluate the prognostic and clinicopathological value of EPHA1 expression. The I2 value was used to evaluate the heterogeneity among the included studies. If the I2 value is 50% or more, random-effect model was selected, otherwise, fixed-effect model was applied. Funnel plots and Egger’s regression tests were also conducted to show publication bias. Sensitivity analysis was performed to show the reliability of the pooled results. All of the data were analyzed using StataSE12 (Stata, College Station, TX, USA), and it was determined to be statistically significant if the P value was less than 0.05.
Results
Characteristics of the included studies
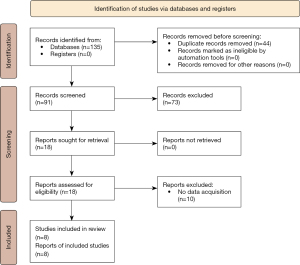
Of the 135 studies initially searched, eight were adopted for analysis (Figure 1). The basic information of the included studies is summarized in Table 1. The included studies comprised of 1,079 patients with different cancers including gastric cancer (n=4), epithelial ovarian cancer (n=1), clear cell renal cell carcinoma (n=1), non-small cell lung cancer (n=1), and colorectal cancer (n=1).
Table 1
| Study | Country | Cancer type | Sample size | Sex of patients (male/female) | Study period | Follow-up (months) | Survival outcome | EPHA1 detection | Cut-off value of EPHA1 expression | Survival analysis | NOS |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Wang et al. (2020) | China | Gastric cancer | 57 | 37/20 | 2015–2016 | Till Oct 2019 | OS | IHC | Staining scores with proportion (≥2, 25–50%) | MVA | 8 |
| Nagare et al. (2020) | India | Epithelial ovarian cancer | 101 | – | 2005–2007 | Median 48.8 | OS, EFS | IHC | Staining scores with intensity and proportion (≥3) | UVA | 7 |
| Inokuchi et al. (2018) | Japan | Gastric cancer | 114 | 89/25 | 2003–2007 | Median 60 | DSS, RFS | IHC | Staining scores with intensity and proportion (≥3) | MVA | 8 |
| Nakagawa et al. (2015) | Japan | Gastric cancer | 222 | 168/54 | 2003–2007 | Mean 58.4 | RFS | IHC | Staining scores with intensity and proportion (≥4) | MVA | 8 |
| Toma et al. (2014) | Germany | Clear cell renal cell carcinoma | 241 | 160/81 | 1993–2006 | NA | PFS, TSS, OS | IHC | Weakly and moderately positive | MVA | 7 |
| Giaginis et al. (2014) | Greece | Non-small cell lung cancer | 88 | 72/16 | NA | Mean 25.39 | OS | IHC | Staining scores with intensity and proportion (≥3) | UVA | 7 |
| Wang et al. (2010) | China | Gastric cancer | 145 | 105/40 | 2002–2006 | Median 24 | OS | IHC | Staining scores with intensity and proportion (by comparing the scores of tumor tissues and adjacent normal tissues) | SC | 7 |
| Dong et al. (2009) | China | Colorectal cancer | 111 | 66/45 | 2004–2006 | Median 25 | OS | IHC | Staining scores with intensity and percentage (by comparing the scores of tumor tissues and matched normal tissues) | SC | 7 |
EPHA1, ephrin receptor A1; NOS, Newcastle-Ottawa Scale; OS, overall survival; EFS, event-free survival; DSS, disease-specific survival; RFS, relapse-free survival; PFS, progression-free survival; TSS, tumor-specific survival; IHC, immunohistochemistry; MVA, multivariate analysis; UVA, univariate analysis; SC, survival curve; NA, not available.
Association between EPHA1 expression and overall survival (OS)
The analysis of the association between EPHA1 expression and OS, disease-specific survival, or tumor-specific survival included 7 studies with 814 cancer patients. In this meta-analysis, disease-specific or tumor-specific survival was regarded as OS.
The included studies were evaluated using a random-effects model due to the severe heterogeneity (I2=80.2%, P<0.001) present among the included studies. The pooled HR for the association between EPHA1 expression and OS was 1.39 (95% CI: 0.79–2.44, P=0.249) (Figure 2A). Subgroup analysis revealed that EPHA1 expression was related to poor OS in patients with gastric cancer (HR 2.23, 95% CI: 1.42–3.51, P<0.001) (Figure 2B).

Association between EPHA1 expression and progression-free survival (PFS)
The analysis of the association between EPHA1 expression and PFS, relapse-free survival, or event-free survival included 4 studies with 678 patients with cancer. In this meta-analysis, relapse-free or event-free survival was considered as PFS.
The included studies were assessed using a fixed-effects model due to the low heterogeneity (I2=0.0%, P=0.584) present among the included studies. The pooled HR was 1.79 (95% CI: 1.49–2.15, P<0.001), indicating that EPHA1 expression is related to the disease progression in cancer patients (Figure 3A). Moreover, in the subgroup analysis between cancer types, the gastric cancer group and “others” cancer group still showed significant results (gastric cancer, HR 1.87, 95% CI: 1.53–2.30, P<0.001; Others, HR 1.50, 95% CI: 1.01–2.24, P=0.047) (Figure 3B).

Association between EPHA1 expression and clinicopathological factors
When analyzed with various cancers, there were no significant results obtained from the analysis of EPHA1 expression and clinicopathological factors (Table 2). However, EPHA1 expression was significantly associated with higher tumor stage (OR 1.74, 95% CI: 1.15–2.61, P=0.008) and lymph node metastasis (OR 1.88, 95% CI: 1.24–2.87, P=0.003) (Table 3, Figure 4A,4B) in gastric cancer.
Table 2
| Characteristic | Number of studies | Number of patients | Pooled OR (95% CI) | P value | Heterogeneity | ||
|---|---|---|---|---|---|---|---|
| I2 (%) | P value | Model | |||||
| Age (old vs. young) | 6 | 781 | 1.17 (0.85–1.62) | 0.342 | 0.0 | 0.513 | Fixed |
| Sex of patients (male vs. female) | 5 | 680 | 0.95 (0.65–1.39) | 0.799 | 14.7 | 0.320 | Fixed |
| Tumor grade (high vs. low) | 7 | 1022 | 1.21 (0.63–2.33) | 0.561 | 80.1 | <0.001 | Random |
| Tumor stage (high vs. low) | 6 | 921 | 1.16 (0.58–2.33) | 0.675 | 78.1 | <0.001 | Random |
| Lymph node metastasis (present vs. absent) | 6 | 921 | 1.28 (0.62–2.65) | 0.499 | 75.2 | 0.001 | Random |
| TNM stage (high vs. low) | 4 | 579 | 1.85 (0.56–6.09) | 0.314 | 86.6 | <0.001 | Random |
OR, odds ratio; CI, confidence interval; EPHA1, ephrin receptor A1; TNM, tumor-node-metastasis.
Table 3
| Characteristic | Number of studies | Number of patients | Pooled OR (95% CI) | P value | Heterogeneity | ||
|---|---|---|---|---|---|---|---|
| I2 (%) | P value | Model | |||||
| Age (old vs. young) | 3 | 481 | 1.02 (0.69–1.52) | 0.917 | 0.0 | 0.804 | Fixed |
| Sex of patients (male vs. female) | 3 | 481 | 1.15 (0.74–1.78) | 0.539 | 0.0 | 0.441 | Fixed |
| Tumor grade (high vs. low) | 3 | 481 | 1.83 (0.80–4.17) | 0.153 | 76.6 | 0.014 | Random |
| Tumor stage (high vs. low) | 3 | 481 | 1.74 (1.15–2.61) | 0.008 | 35.5 | 0.212 | Fixed |
| Lymph node metastasis (present vs. absent) | 3 | 481 | 1.88 (1.24–2.87) | 0.003 | 26.8 | 0.255 | Fixed |
| TNM stage (high vs. low) | 2 | 367 | 4.98 (1.00–24.91) | 0.051 | 82.2 | 0.018 | Random |
EPHA1, ephrin receptor A1; OR, odds ratio; CI, confidence interval; TNM, tumor-node-metastasis.

Publication bias
Funnel plots were created, and the results showed an asymmetric distribution (Figure 5A,5B). However, Egger’s regression test did not show a small-study effect (for OS, P=0.275; for PFS, P=0.501). In addition, we performed filled funnel plots, and the results for OS revealed that the HR was reduced and the range of 95% CI was downgraded (HR 1.05, 95% CI: 0.60–1.82, P=0.867) compared with the initial pooled results (Figure 5C). The results for PFS showed that data did not change (Figure 5D).

Sensitivity analysis
For OS, it was confirmed that the study published by Nagare et al. (5) had a significant effect on the initial pooled results (HR 1.64, 95% CI: 1.24–2.17) (Figure 6A). The pooled results of the sensitivity analysis showed that the HR was decreased and the range of 95% CI was narrowed (HR 1.14, 95% CI: 0.91–1.43) as compared with the initial pooled results (Figure 6A).

For PFS, the overall results were the same as the initial pooled results (HR 1.79, 95% CI: 1.49–2.15), although the study published by Nakagawa et al. (6) seemed to have an impact (HR 1.63, 95% CI: 1.16–2.29) (Figure 6B).
Discussion
EPHA1 is a component of the ephrin receptor, which belongs to the receptor tyrosine kinase family (13). EPHA1 is the first member of the ephrin receptor and it is located on chromosome 7q34 (14). EPHA1 is expressed in various normal tissues such as the intestine, lung, kidney, bladder, and thymus. It is also seen in cancer cells, including colorectal carcinoma, gastric cancer, lung cancer, ovarian carcinoma, renal cell carcinoma, and head and neck squamous cell carcinoma (5-10,12-15). Recent studies have reported that EPHA1 is carcinogenic through interaction with tumor cells as well as interaction with the surrounding tumor microenvironment, and it could also be a potential therapeutic target (10,13,15). Moreover, EPHA1 is known to be closely related to the survival of cancer patients (5-12). Thus, we systematically analyzed the effects of EPHA1 on the survival of cancer patients.
In the present study, we identified eight eligible studies consisting of 1079 patients with cancer to evaluate the association between survival and EPHA1 expression in patients with cancer. Wang et al. (10,11) and Inokuchi et al. (8) demonstrated that EPHA1 expression was correlated with poor OS in patients with gastric cancer. Toma et al. (9) reported the association between EPHA1 expression and unfavorable OS in clear cell renal cell carcinoma. Nagare et al. (5), Giaginis et al. (7), and Dong et al. (12) showed that there was no significant clear link between EPHA1 expression and OS in epithelial ovarian cancer, non-small cell lung cancer, and colorectal cancer, respectively. With respect to PFS, Nagare et al. (5), Inokuchi et al. (6), Nakagawa et al. (6), and Toma et al. (9) reported consistent results that EPHA1 expression was closely related to PFS of cancer patients.
In this study, we demonstrated that EPHA1 expression is associated with poor OS in patients with gastric cancer and is related to PFS in patients with cancer. We also found that EPHA1 expression is correlated with higher tumor stage and lymph node metastasis in gastric cancer.
However, this study has a limitation in that the number of small samples and the heterogeneity between the included studies have not been overcome. We hope that further research will be carried out.
In summary, we systematically evaluated the relationship between EPHA1 expression and survival in patients with cancer for the first time. We revealed that EPHA1 expression was significantly associated with PFS in patients with cancer.
Acknowledgments
Funding: None.
Footnote
Reporting Checklist: The authors have completed the PRISMA reporting checklist. Available at https://tcr.amegroups.com/article/view/10.21037/tcr-21-1367/rc
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://tcr.amegroups.com/article/view/10.21037/tcr-21-1367/coif). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Pergaris A, Danas E, Goutas D, et al. The Clinical Impact of the EPH/Ephrin System in Cancer: Unwinding the Thread. Int J Mol Sci 2021;22:8412. [Crossref] [PubMed]
- Ieguchi K, Maru Y. Roles of EphA1/A2 and ephrin-A1 in cancer. Cancer Sci 2019;110:841-8. [Crossref] [PubMed]
- Lisle JE, Mertens-Walker I, Rutkowski R, et al. Eph receptors and their ligands: promising molecular biomarkers and therapeutic targets in prostate cancer. Biochim Biophys Acta 2013;1835:243-57. [PubMed]
- Jin Y, Zou Y, Wan L, et al. Decreased Eph receptor A1 expression is related to grade in ovarian serous carcinoma. Mol Med Rep 2018;17:5409-15. [Crossref] [PubMed]
- Nagare RP, Sneha S, Sidhanth C, et al. Expression of cancer stem cell markers CD24, EPHA1 and CD9 and their correlation with clinical outcome in epithelial ovarian tumours. Cancer Biomark 2020;28:397-408. [Crossref] [PubMed]
- Nakagawa M, Inokuchi M, Takagi Y, et al. Erythropoietin-Producing Hepatocellular A1 is an Independent Prognostic Factor for Gastric Cancer. Ann Surg Oncol 2015;22:2329-35. [Crossref] [PubMed]
- Giaginis C, Tsoukalas N, Bournakis E, et al. Ephrin (Eph) receptor A1, A4, A5 and A7 expression in human non-small cell lung carcinoma: associations with clinicopathological parameters, tumor proliferative capacity and patients' survival. BMC Clin Pathol 2014;14:8. [Crossref] [PubMed]
- Inokuchi M, Nakagawa M, Baogok N, et al. Prognostic Significance of High EphA1-4 Expression Levels in Locally Advanced Gastric Cancer. Anticancer Res 2018;38:1685-93. [PubMed]
- Toma MI, Erdmann K, Diezel M, et al. Lack of ephrin receptor A1 is a favorable independent prognostic factor in clear cell renal cell carcinoma. PLoS One 2014;9:e102262. [Crossref] [PubMed]
- Wang YC, Dai Y, Xu GL, et al. Association Between EphA1 and Tumor Microenvironment in Gastric Carcinoma and its Clinical Significance. Med Sci Monit 2020;26:e923409. [Crossref] [PubMed]
- Wang J, Dong Y, Wang X, et al. Expression of EphA1 in gastric carcinomas is associated with metastasis and survival. Oncol Rep 2010;24:1577-84. [PubMed]
- Dong Y, Wang J, Sheng Z, et al. Downregulation of EphA1 in colorectal carcinomas correlates with invasion and metastasis. Mod Pathol 2009;22:151-60. [Crossref] [PubMed]
- Theocharis S, Klijanienko J, Giaginis C, et al. Ephrin receptor (Eph) -A1, -A2, -A4 and -A7 expression in mobile tongue squamous cell carcinoma: associations with clinicopathological parameters and patients survival. Pathol Oncol Res 2014;20:277-84. [Crossref] [PubMed]
- Wang X, Liu Y, Cao G, et al. Expression of the EphA1 protein is associated with Fuhrman nuclear grade in clear cell renal cell carcinomas. Int J Clin Exp Pathol 2015;8:6821-7. [PubMed]
- Giaginis C, Tsourouflis G, Zizi-Serbetzoglou A, et al. Clinical significance of ephrin (eph)-A1, -A2, -a4, -a5 and -a7 receptors in pancreatic ductal adenocarcinoma. Pathol Oncol Res 2010;16:267-76. [Crossref] [PubMed]