Diagnostic test of dynamic computed tomography in early gastrointestinal lymphoma and precancerous lesions
Introduction
There are two types of malignant lymphoma: malignant tumors of the lymph nodes and malignant tumors of tissues beyond the lymph nodes. It is a malignant tumor mainly of lymphocytes in the body (1). Lymphoma can occur at any age, but mostly in middle and young age and there are more male patients than female patients (incidence ratio ≈3:1) (2). In China, malignant lymphoma ranks 8th among new cases of all tumors and 10th among new cases of female tumors, with an incidence of about 6–7 cases per 100,000 (3). In clinical pathology, lymphoma is generally divided into Hodgkin’s lymphoma and non-Hodgkin’s lymphoma (NHL), the latter being one of the cancers with the fastest increasing incidence, accounting for >90%. To date, the etiology and pathogenesis of lymphoma are not clear, and most researchers consider that it is related to psychological stress, blood transfusion, viral infection, immune suppression caused by the treatment of other diseases such as organ transplantation and industrial environmental pollution (4,5).
Gastrointestinal lymphoma originates from the gastrointestinal tract, and includes gastric and small intestine lymphoma and immune deficiency-associated lymphoma. It is relatively rare; most cases are NHL, and B-cell lymphoma is common (6). Among gastrointestinal lymphoma, the highest incidence rate is gastric lymphoma, followed by small intestinal lymphoma and colon lymphoma. The specific pathogenesis is unclear at present, but some experts speculate that it is related to interaction of internal and external factors that disturbs homeostasis, resulting in abnormal proliferation and differentiation of lymphocytes (7). Several findings have led to this speculation. (I) It has been associated with bacterial infections such as Helicobacter pylori (HP) (8). (II) Chromosomal aberrations (structural aberration or chromosome number abnormality such as deletion, translocation, and insertion) are identified in >90% of patients with lymphoma (9). (III) The incidence of gastrointestinal lymphoma is increased by the long-term effects of physical and chemical factors or high doses of radiation (10). (IV) Retroviruses can infect T lymphocytes in human body, and the acquired immune deficiency syndrome (AIDS) virus is a retrovirus. About 5–10% of AIDS patients develop lymphoma, especially of the small intestine (11). (V) Primary lymphoma of the small intestine is caused by many diseases, as well as immune dysfunction (12).
It is well known that the occurrence and development of malignant tumors is a long and complex process from normal epithelial cells to simple hyperplasia to atypical hyperplasia to carcinoma in situ to invasive cancer, precipitated by carcinogenic factors. The scientific consensus is that precancerous lesions begin with atypical hyperplasia (13), but the occurrence and development of cancer is not inevitable. Nevertheless, the vast majority of cancers have obvious precancerous lesions. Thus, identifying precancerous lesions at the early stage of the disease is a very valuable opportunity for early diagnosis and treatment (14,15).
With medical endoscopy becoming more commonly performed in recent years, precancerous lesions of the digestive system and early-stage lymphoma have attracted the attention of the medical community (16). If accurate early diagnosis can be made and the most effective early treatment is implemented, the survival rate of patients will be remarkably increased and their quality of life improved. Complete cure can be achieved (17).
Dynamic computed tomography (CT) scan is a method of scanning after intravenous injection of water-soluble contrast agent, which can improve the density difference between the lesion and adjacent tissues to reflect the lesion and improve the display rate of the lesion. It has been widely used in recent years, especially in the differential diagnosis of lymphoma, which has obvious advantages over conventional imaging (18). Dynamic enhanced CT is used by clinicians to make a preliminary judgment of pathological changes and then effective treatment measures are implemented (19,20).
In this study, we used dynamic enhanced CT to explore its diagnostic value in differentiating early gastrointestinal lymphoma from precancerous lesions, thus providing a theoretical basis for the differential diagnosis of these two diseases in clinical practice. We present the following article in accordance with the STARD reporting checklist (available at https://tcr.amegroups.com/article/view/10.21037/tcr-22-1085/rc).
Methods
General data
The study group comprised 82 patients with gastrointestinal lesions admitted to the Department of Gastroenterology of Huizhou Municipal Central People’s Hospital from May 2018 to May 2021. The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). The study was approved by ethics board of Huizhou Municipal Central People’s Hospital (No. kyll20210108) and informed consent was taken from all the patients. All patients complained of long-term abdominal discomfort.
The inclusion criteria were: age between 16 and 82 years; recurrent abdominal discomfort (abdominal pain, abdominal distention), hiccups, acid reflux, loss of appetite, body fatigue, and other symptoms; all relevant clinical data available; suspicious lesions found by routine gastroscopy, such as abnormal morphology on the surface of gastric mucosa or abnormal morphology of microvessels or even disappearance, clear lesions, mucosal congestion, swelling, granular appearance, and erosion, consistent with relevant diagnostic and treatment guidelines (21).
The exclusion criteria were: coagulation dysfunction; unable to tolerate the examination method; digestive tract obstruction or esophageal varices; respiratory tract infection within the past 3 months; serious dysfunction of heart, liver, spleen, kidney, lung, or other organs; pregnancy or lactation; history of radical treatment of HP infection or a history of gastric surgery; participation in other clinical trials simultaneously; history of taking nonsteroidal anti-inflammatory drugs in the past 6 months; abnormal mental or cognitive function; advanced gastric cancer, gastric polyps, or gastric bleeding diagnosed by gastroscopy.
CT scanning
All patients underwent CT examination by a 64-slice spiral CT scanner Lightspeed VCT (GE Company, Boston, USA). Patients were asked to fast for 4–6 h and drink 800–1,200 mL of water 45 min before the scan, as well as another 250 mL of water when entering the CT room. The patients lay supine during the scan. An ANT200200 CT double-cylinder high-pressure syringe (Antecedent Medical Co., Ltd., Shenzhen, China) was used to inject 85–110 mL of contrast agent (ioversol 320, iodine content 1 mL:320 mg I) through the median cubital vein at a flow rate of 2.0 mL/s. The scanning delay times were 30 and 50 s. Dynamic enhanced CT scanning was implemented in the arterial and portal vein phases, which required patients to hold their breath. The scanning range was from the upper diaphragm to the lower margin of both kidneys, with the following parameters: scanning layer thickness 5.0 mm, scanning layer spacing 5.0 mm, tube voltage 120 kV, and tube current 220 mA.
CT imaging analysis
The final analysis of the CT images was performed by two experienced radiologists: a resident with 5 years of clinical experience, and an associate chief physician with 12 years of clinical experience. The size of the region of interest (ROI) in the center of the lesion was determined and measured in the uniform density of the lesion, about 8–22 mm2. Areas of necrosis and vascular structures were not outlined together. At the same time, the CT values of the plain scan, the arterial phase, and the portal venous phase areas of the ROI of each lesion were measured. The average value was calculated from the results of the three measurements.
Observation indexes
The following indexes were recorded: (I) comparison of the general data of the patients; (II) comparison of lesion sites between the two groups of patients; (III) comparison of thickness of the affected gastric wall between the two groups; (IV) comparison of the density difference of the arterial phase and plain scan CT (δ1) and the density ratio of the arterial phase and plain scan CT (Q1) between the two groups; (V) comparison of the density difference of the portal vein phase and plain scan CT (δ2) and the density ratio of portal vein phase and plain scan CT (Q2) between the two groups.
Statistical analysis
SPSS 20.0 was used for statistical data analysis. Measurement data are expressed as mean ± standard deviation and enumeration data are represented by number. The measurement data were in accordance with a normal distribution. Independent t-test was used to test the difference of parameter characteristics between the two groups of patients undergoing dynamic enhanced CT. Taking pathological diagnosis results as the gold standard, receiver operating characteristic (ROC) curve of different dynamic contrast-enhanced CT scanning parameters was drawn, the area under ROC curve (AUC) was calculated, and the maximum matching value of Youden Index (YI) was taken as the diagnosis threshold. The difference was statistically significant at P<0.05.
Results
General data of the patients
The 82 patients with gastrointestinal lesions were classified as early lymphoma (n=32) and precancerous lesion (n=50) groups based on the type of lesion. Figures 1,2 respectively show the general data and CT images of the patients in the two groups after admission.


Comparison of lesion sites and the affected gastric wall thickness in the two groups
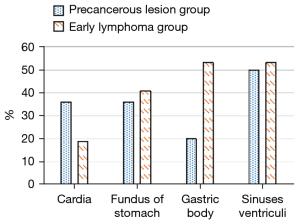
The most commonly involved sites of early lymphoma were the gastric antrum and gastric body, whereas the most commonly involved sites of precancerous lesions were the gastric antrum, followed by the cardia and gastric fundus. There were 21 patients (42.0%) with precancerous lesions and 15 patients (46.9%) with early lymphoma. There was no statistically significant difference in lesion sites between the two groups (P>0.05) (Figure 3).
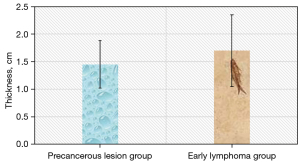
The gastric wall thickness in the early lymphoma group was 0.2–3.1 cm, with an average thickness of 1.70±0.65 cm. In the precancerous lesion group, the wall was 0.3–3.1 cm, with an average thickness of 1.45±0.43 cm. There was no significant difference in the affected gastric wall thickness between the two groups (P>0.05) (Figure 4).
Comparison of δ1 and Q1 of the arterial phase and plain scan CT between the two groups
The values of δ1 and Q1 were compared between groups. The respective mean values of δ1 in the early lymphoma and precancerous lesion groups were 14.57±6.86 and 17.28±7.61 Hounsfield units (HU), the maximum values were 24.00 and 36.88 HU, and the minimum values were 4.00 and 2.72 HU. There was no significant difference in the δ1 values between the two groups (P>0.05). The respective mean values of Q1 in the early lymphoma and precancerous lesion groups were 1.30±0.21 and 1.40±0.16, the maximum values were 1.60 and 2.35, and the minimum values were 1.01 and 1.07. There was no significant difference in Q1 between groups (P>0.05) (Figures 5,6).


Comparison of the density difference δ2 and ratio Q2 of the portal phase and plain scan CT between the two groups
The values of δ2 and Q2 were compared between the two groups. The respective mean values of δ2 in the early lymphoma and precancerous lesion groups were 29.50±6.05 and 41.55±10.10 HU, the maximum values were 44.70 and 70.40 HU, and the minimum values were 17.00 and 21.83 HU. There was significant difference between the two groups (P<0.05). The respective mean values of Q2 in the early lymphoma and precancerous lesion groups were 1.70±0.05 and 2.06±0.31, the maximum values were 2.12 and 3.02, and the minimum values were 1.33 and 1.46. There was a significant difference in Q2 between the two groups (P<0.05) (Figures 7,8).


Calculation of ROC curve
The AUC value of the maximum area under the ROC curve of early lymphoma and precancerous lesions was 0.755 using the density difference of the portal venous phase and plain scan CT. When δ2 =35.63 HU, the specificity was 89.60%, sensitivity was 67.50%, and the YI was 0.498. According to the density ratio Q2 of the portal venous phase and plain scan CT, the AUC of the maximum area under the ROC curve of early lymphoma and precancerous lesions was 0.878. When Q2 =1.86, the specificity was 89.60%, sensitivity was 64.90%, and the YI was 0.472. Cases in the two groups that were consistent with δ2 ≥35.63 HU and Q2 ≥1.86 were further obtained. After the calculation, the diagnostic specificity of the combination of the two indicators was 98.99% and the sensitivity was 56.80% (Figure 9).

Discussion
There is an integral connection between the histological structure of early gastrointestinal lymphoma and precancerous lesions detected by dynamic enhanced CT; that is, precancerous lesions usually occur in the gastrointestinal tract mucosa, muscle fiber and epithelium. During pathological change, many vascular endothelial growth factors are released, which contribute to neo-angiogenesis. The CT manifestation of contrast agent retention at the venous stage is continuous enhancement (22,23). Lymphoma is a solid tumor originating in the lamina propria of the gastric mucosa and the lymphocytic follicular cells of the submucosa. The dense arrangement of tumor cells leads to poor blood supply, so the degree of enhancement is not as high as in precancerous lesions (24). The significant histological differences in blood supply are reflected in the dynamic enhanced CT images. However, the CT perfusion or dynamic enhanced CT examination is time-consuming, and the corresponding diagnostic images are obtained later from the workstation. So even though both CT perfusion and dynamic enhanced CT examination can provide hemodynamic information for the differential diagnosis of early lymphoma from precancerous lesions, they are not performed widely in the daily practice (25).
Currently, there are few studies related to dynamic enhanced CT for early gastrointestinal lymphoma diagnosis. Ma et al. (26) showed that the density difference δ1 of the arterial phase and plain scan CT in early gastric lymphoma and the density difference δ2 of the portal phase and plain scan CT were increased by ≈8 and ≈18 HU on average, respectively. Moreover, the δ1 and δ2 of precancerous lesions were increased by 18 and 38 HU on average, respectively. In this study, the values of both are refined, and on this basis, the ratio Q between arterial phase, portal vein phase and plain CT density is introduced. We used ROC curve analysis to find the optimal threshold for differentiating early lymphoma from precancerous lesions. There was no significant difference between δ1 and Q1 (P>0.05), but a significant difference between δ2 and Q2 (P>0.05), which could be related to the vascular distribution of the two diseases. The blood supply of precancerous lesions is good, but the CT enhancement in the arterial phase is not obvious because most are capillaries. However, obvious enhancement occurred after contrast agent filling in the venous phase or delayed phase. In contrast, the poor blood supply of early-stage lymphoma showed the difference in enhancement mainly in the venous phase.
By analyzing the calculation results of the ROC curves, when δ2 ≥35.63 HU and Q2 ≥1.86, precancerous lesions were more likely to be diagnosed. The specificity was 89.60% and 67.50%, respectively, and the sensitivity was 89.60% and 64.90%, respectively, which indicated that the diagnosis efficiency of δ2 was similar to that of Q2. However, both had high sensitivity and specificity. Therefore, they can be used as effective indexes to differentiate early lymphoma from precancerous lesions. Further analysis with δ2 and Q2 showed that the diagnostic specificity of the two indexes combined was 98.99%, and the sensitivity was 56.80%. Although the sensitivity decreased, the specificity increased markedly, which indicated that when δ2 ≥35.63 HU and Q2 ≥1.86, dynamic enhanced CT could be used to diagnose precancerous lesions. This conclusion is similar to that of Niu et al. (27).
Conclusions
We explored using dynamic enhanced CT scan for differential diagnosis of early gastrointestinal lymphoma and precancerous lesions. Our results showed that when the two indexes, δ2 and Q2, were combined, the specificity and sensitivity of diagnosis were 98.99% and 56.80%, respectively. Therefore, dynamic enhanced CT and Q2 effectively differentiated early gastrointestinal lymphoma from precancerous lesions, and improved diagnostic accuracy. However, the sample in this study was relatively small, so it needs to be expanded in the future clinical trials.
Acknowledgments
Funding: This study was supported by
Footnote
Reporting Checklist: The authors have completed the STARD reporting checklist. Available at https://tcr.amegroups.com/article/view/10.21037/tcr-22-1085/rc
Data Sharing Statement: Available at https://tcr.amegroups.com/article/view/10.21037/tcr-22-1085/dss
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://tcr.amegroups.com/article/view/10.21037/tcr-22-1085/coif). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). The study was approved by ethics board of Huizhou Municipal Central People’s Hospital (No. kyll20210108) and informed consent was taken from all the patients.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Ohmoto A, Fuji S. Histological transformation in malignant lymphoma: a possible role of PET/CT and circulating tumor DNA as noninvasive diagnostic tools. Expert Rev Hematol 2020;13:23-30. [Crossref] [PubMed]
- Jiang M, Bennani NN, Feldman AL. Lymphoma classification update: T-cell lymphomas, Hodgkin lymphomas, and histiocytic/dendritic cell neoplasms. Expert Rev Hematol 2017;10:239-49. [Crossref] [PubMed]
- Buske C, Hutchings M, Ladetto M, et al. ESMO Consensus Conference on malignant lymphoma: general perspectives and recommendations for the clinical management of the elderly patient with malignant lymphoma. Ann Oncol 2018;29:544-62. [Crossref] [PubMed]
- McCarten KM, Nadel HR, Shulkin BL, et al. Imaging for diagnosis, staging and response assessment of Hodgkin lymphoma and non-Hodgkin lymphoma. Pediatr Radiol 2019;49:1545-64. [Crossref] [PubMed]
- Abdelwahed Hussein MR. Non-Hodgkin's lymphoma of the oral cavity and maxillofacial region: a pathologist viewpoint. Expert Rev Hematol 2018;11:737-48. [Crossref] [PubMed]
- Kono Y, Kusumoto C, Kiguchi T, et al. Gastrointestinal: Rapid emergence of double-expressor lymphoma after Helicobacter pylori eradication therapy. J Gastroenterol Hepatol 2021;36:299. [Crossref] [PubMed]
- Lane J, Price J, Moore A, et al. Low-grade gastrointestinal lymphoma in dogs: 20 cases (2010 to 2016). J Small Anim Pract 2018;59:147-53. [Crossref] [PubMed]
- Yeo SH, Jung K, Park MI, et al. Mucosa-associated Lymphoid Tissue Lymphoma-mimicking Primary Gastrointestinal Small Lymphocytic Lymphoma. Korean J Gastroenterol 2020;75:212-5. [Crossref] [PubMed]
- Saito M, Suzuki H, Kono K, et al. Treatment of lung adenocarcinoma by molecular-targeted therapy and immunotherapy. Surg Today 2018;48:1-8. [Crossref] [PubMed]
- Mwazha A, Nhlonzi GB, Mazengenya P. Gastrointestinal Tract Plasmablastic Lymphoma in HIV-Infected Adults: A Histopathological Review. Int J Surg Pathol 2020;28:735-48. [Crossref] [PubMed]
- Lei GY, Tay KV, Teo LT. Rare primary B-cell lymphoma of small intestine presenting as giant jejunal diverticulum and complicated by phytobezoar and intestinal obstruction. ANZ J Surg 2021;91:E238-9. [Crossref] [PubMed]
- Cheng J, Guo J, North BJ, et al. Functional analysis of deubiquitylating enzymes in tumorigenesis and development. Biochim Biophys Acta Rev Cancer 2019;1872:188312. [Crossref] [PubMed]
- Zheng C, Jia HY, Liu LY, et al. Molecular fingerprint of precancerous lesions in breast atypical hyperplasia. J Int Med Res 2020;48:300060520931616. [Crossref] [PubMed]
- Li ZW, Zhou SH. Laryngopharyngeal reflux disease and laryngeal precancerous lesions. Zhonghua Er Bi Yan Hou Tou Jing Wai Ke Za Zhi 2017;52:637-9. [PubMed]
- Miyazaki T, Shinkawa H, Takemura S, et al. Precancerous Lesions and Liver Atrophy as Risk Factors for Hepatolithiasis-Related Death after Liver Resection for Hepatolithiasis. Asian Pac J Cancer Prev 2020;21:3647-54. [Crossref] [PubMed]
- Zhang Y, Zhou P. Value of endoscopy application in the management of complications after radical gastrectomy for gastric cancer. Zhonghua Wei Chang Wai Ke Za Zhi 2017;20:160-5. [PubMed]
- Guo L, Xiao X, Wu C, et al. Real-time automated diagnosis of precancerous lesions and early esophageal squamous cell carcinoma using a deep learning model (with videos). Gastrointest Endosc 2020;91:41-51. [Crossref] [PubMed]
- Hyodo R, Takehara Y, Nishida A, et al. "Speckled Enhancement" on Gd-EOB-DTPA Enhanced MR Imaging of Primary Hepatic Mucosa-associated Lymphoid Tissue Lymphoma. Magn Reson Med Sci 2021; Epub ahead of print. [Crossref] [PubMed]
- Ozaki K, Ikeno H, Koneri K, et al. Primary hepatic diffuse large B-cell lymphoma presenting unusual imaging features. Clin J Gastroenterol 2020;13:1265-72. [Crossref] [PubMed]
- Örgüç S, Arkun R. Tumor-like Lesions of Bone and Soft Tissues and Imaging Tips for Differential Diagnosis. Semin Musculoskelet Radiol 2020;24:613-26. [Crossref] [PubMed]
- Huynh DK, Toscano L, Phan VA, et al. Ultrathin disposable gastroscope for screening and surveillance of gastroesophageal varices in patients with liver cirrhosis: a prospective comparative study. Gastrointest Endosc 2017;85:1212-7. [Crossref] [PubMed]
- Bakhshi S, Bhethanabhotla S, Kumar R, et al. Posttreatment PET/CT Rather Than Interim PET/CT Using Deauville Criteria Predicts Outcome in Pediatric Hodgkin Lymphoma: A Prospective Study Comparing PET/CT with Conventional Imaging. J Nucl Med 2017;58:577-83. [Crossref] [PubMed]
- Liu XH, Jin F, Zhang M, et al. Peripheral T cell lymphoma after chronic lymphocytic inflammation with pontine perivascular enhancement responsive to steroids (CLIPPERS): a case report. BMC Neurol 2019;19:266. [Crossref] [PubMed]
- Goel Y, Yadav S, Pandey SK, et al. Tumor Decelerating and Chemo-Potentiating Action of Methyl Jasmonate on a T Cell Lymphoma In Vivo: Role of Altered Regulation of Metabolism, Cell Survival, Drug Resistance, and Intratumoral Blood Flow. Front Oncol 2021;11:619351. [Crossref] [PubMed]
- Zou M, Zhao Z, Zhang B, et al. Pulmonary lesions: correlative study of dynamic triple-phase enhanced CT perfusion imaging with tumor angiogenesis and vascular endothelial growth factor expression. BMC Med Imaging 2021;21:158. [Crossref] [PubMed]
- Ma Z, Fang M, Huang Y, et al. CT-based radiomics signature for differentiating Borrmann type IV gastric cancer from primary gastric lymphoma. Eur J Radiol 2017;91:142-7. [Crossref] [PubMed]
- Niu X, Jiang W, Zhang X, et al. Comparison of Contrast-Enhanced Ultrasound and Positron Emission Tomography/Computed Tomography (PET/CT) in Lymphoma. Med Sci Monit 2018;24:5558-65. [Crossref] [PubMed]
(English Language Editor: K. Brown)