Metronomic chemotherapy combined with immunotherapy in solid tumor: a systematic review
Highlight box
Key findings
• The research indicates that combining metronomic chemotherapy (MC) with immune checkpoint inhibitors (ICIs) enhances treatment outcomes in solid tumors like non-small cell lung cancer, ovarian cancer, and breast cancer. This approach is particularly beneficial for patients with poor responses to standard treatments. Efficacy depends on factors such as programmed cell death ligand 1 (PD-L1) expression, epidermal growth factor receptor (EGFR) mutation, tumor subtype, and treatment stage, with better results seen in patients with PD-L1 ≥1% and those treated earlier. The combination is generally well-tolerated, with most side effects being mild, while severe side effects are rare and manageable.
What is known and what is new?
• MC modulates the immune microenvironment by inhibiting regulatory T cells (Tregs)/myeloid-derived suppressor cells and activating dendritic cells (DCs), with favorable safety. ICIs boost antitumor immunity but have limited efficacy as monotherapy in some tumors. The combination of MC and ICI synergistically enhances immune activation, reduces nutritional competition, and aids immune cell infiltration in various solid tumors. This approach benefits patients with driver gene mutations like EGFR or those resistant to osimertinib, and triplet regimens (MC + ICI + bevacizumab) demonstrate promise in recurrent ovarian cancer. Additionally, metronomic dosing of vinorelbine/cyclophosphamide works better with ICIs than conventional chemotherapy in certain subtypes like squamous cell lung cancer.
What is the implication, and what should change now?
• MC + ICI offers a less toxic and effective treatment for advanced solid tumor patients who do not respond to standard therapies, broadening treatment options and enhancing quality of life. To confirm efficacy, it is suggested to conduct large-scale phase III trials; create composite biomarkers (PD-L1, tumor mutational burden, homologous recombination deficiency) for patient stratification; refine regimens (drug choice, dosing, sequencing); investigate first-line/maintenance uses; and use multi-omics to understand synergistic mechanisms for precision medicine.
Introduction
Background
Based on the most recent data analysis conducted by the International Agency for Research on Cancer, which assessed 185 countries across variables such as gender, age, and cancer type, the following five cancers with the highest mortality rates are identified: lung cancer, responsible for 18.7% of global cancer-related deaths in 2022; colorectal cancer, accounting for 9.3%; liver cancer, at 7.8%; female breast cancer, comprising 6.9%; and stomach cancer, representing 6.8% in 2022 (1). According to the 2024 Cancer Statistics Report, there has been a 33% reduction in the combined cancer mortality rate among men and women. This notable achievement is largely attributed to advancements in treatment methodologies and the extensive implementation of early screening programs (2-4). Despite advances in treating solid tumors like lung and breast cancer, advanced-stage prognosis remains poor, especially for those on second-line therapies due to low physical tolerance and high disease progression risk (2). ICIs offer hope but have limited success as monotherapy, with only 10–20% response rates in some cancers due to challenges like immune escape. Metronomic chemotherapy (MC), with low-dose, frequent administration, shows mild toxicity and enhance the immune microenvironment, addressing ICI limitations and supporting their combined use (5-10).
Rationale and knowledge gap
Over the past two decades, substantial advancements have been made in the systemic management of malignant tumors, encompassing modalities such as surgery, radiotherapy, chemotherapy, and targeted therapy. These developments have contributed to enhanced survival rates among patients with diverse cancer types. Nonetheless, the prognosis for individuals with advanced-stage malignant tumors remains unfavorable. This is especially true for patients receiving second-line and subsequent treatments, as their tolerance diminishes, rendering them less capable of withstanding high-intensity chemotherapy. Consequently, these patients are persistently confronted with the risk of disease progression and mortality. Investigating novel therapeutic strategies and regimens for advanced malignant tumors is critically important. MC, distinct from traditional chemotherapy dosing and scheduling, is a treatment modality characterized by the administration of relatively low doses of cytotoxic drugs at frequent intervals in a continuous manner. This approach facilitates the maintenance of a consistently low yet therapeutically effective drug concentration in the bloodstream over an extended duration. Consequently, it has the potential to significantly extend disease control time while markedly reducing toxic side effects (11). Currently, MC has been demonstrated to possess favorable efficacy and safety profiles across various solid tumors (11-14). With the introduction of novel agents such as immune checkpoint inhibitors and an enhanced understanding of the distinct mechanisms underlying MC, combination therapies incorporating MC have attracted significant attention in clinical practice and have prompted numerous innovative investigations. The mechanisms of MC include the inhibition of tumor angiogenesis, direct effects on tumor cells, and modulation of the immune system. Specifically, MC can modulate the immune system by inhibiting regulatory T cells (Tregs) and myeloid-derived suppressor cells (MDSCs), while stimulating dendritic cells (DCs), thereby exerting suppressive effects on tumor cells. Both innate and adaptive immunity are integral to the development and regulation of tumors. MC can affect Treg cells, which have the capacity to suppress antitumor immune responses by inhibiting tumor-specific CD8+ T lymphocytes and CD4+ T helper cells, as well as tumor-nonspecific natural killer (NK) cells and NKT cells. Consequently, the targeted inhibition or depletion of Treg cells to suppress their activity constitutes a strategy for enhancing immune responses against tumor-associated antigens (15,16). MC can further potentiate the host immune response by facilitating the maturation of DCs and inducing apoptosis in tumor cells (7). Cancer immunotherapy, through the activation of immune cells and the enhancement of the body’s antitumor immune response, effectively targets minimal residual disease and inhibits tumor progression. It addresses tumor immune evasion, thereby reactivating immune cells to eradicate cancer cells. Due to its minimal side effects and substantial therapeutic efficacy, cancer immunotherapy has emerged as a promising avenue in cancer treatment and is recognized as the fifth major cancer treatment modality, following surgery, radiotherapy, chemotherapy, and targeted therapy. ICIs are currently the most extensively utilized agents in antitumor immunotherapy. The immunostimulatory properties of MC on tumor immunity make it an ideal adjunct to immunotherapy. Patients with advanced solid tumors face three main challenges: limited treatment options, poor tolerability, and high drug resistance. Combining MC with ICIs offers a synergistic approach: MC reduces tumor angiogenesis and immunosuppressive cells, enhancing the tumor microenvironment (TME) for ICIs. ICIs then boost the anti-tumor immune response by blocking the programmed cell death protein 1/programmed cell death ligand 1 (PD-1/PD-L1) pathway. This dual approach of “microenvironment remodeling + immune activation” may effectively balance efficacy and safety, benefiting patients who do not respond well to standard treatments (17-20).
The clinical use of MC with immunotherapy faces two main challenges: (I) no standardized dosing regimens exist due to variability in drugs, doses, and administration schedules across studies, preventing the establishment of an optimal regimen; (II) patient stratification criteria remain unclear, hindering the identification of those who would benefit most (6,21-25). Additionally, there is a lack of comprehensive reviews on the combination’s effectiveness across different tumor types and its long-term safety. These issues complicate its standardized clinical application, highlighting the need for a systematic review to address these gaps.
Objective
While the synergistic mechanism of MC combined with immunotherapy has been substantiated through basic research (7,25), and numerous early-phase clinical studies (phase I/II, single-arm trials) have demonstrated promising efficacy and safety (8-13), the advancement of large-scale, multicenter phase III clinical trials for this combination regimen has been sluggish. Consequently, there remains a paucity of high-level evidence to endorse its widespread clinical implementation. In this context, a systematic review and qualitative synthesis of the extant data from early-stage clinical studies, alongside a summary of the benefits for patients with various solid tumor types and treatment lines, and an identification of factors influencing efficacy and safety risks, can provide clinicians with timely and critical insights for developing individualized treatment plans for patients exhibiting suboptimal responses to standard therapies. Additionally, this approach can inform the design of subsequent phase III trials. We present this article in accordance with the PRISMA reporting checklist (available at https://tcr.amegroups.com/article/view/10.21037/tcr-2025-1-2912/rc).
Methods
Study selection
The databases of scientific literature, including PubMed, Embase, Cochrane Library, Web of Science, China Wanfang, China CNKI, and China VIP, were searched for relevant articles published as of March 17, 2025. The retrieval process was executed by integrating MeSH terms/Emtree terms with free text words, employing the following specific combination formula: (metronomic OR “metronomic chemotherapy” OR “metronomic dosing”) AND (“immune checkpoint inhibitor” OR “PD-1 inhibitor” OR “PD-L1 inhibitor” OR “CTLA-4 inhibitor”) AND (“solid tumor” OR “ Solid malignancies” OR “solid tumor malignancies”). Two researchers independently conducted literature screening, data extraction and cross-verification, and resolved disagreements through team discussion.
To ensure scientific rigor and standardization in literature screening and inclusion, this systematic review’s scope was precisely defined using the PICOS framework: the Population (P) included patients with confirmed solid tumors; the Intervention (I) was MC with immune checkpoint inhibitors; no Comparison (C) was required, focusing on the regimen’s efficacy and safety; primary Outcomes (O) included median progression-free survival (mPFS), median overall survival (mOS), objective response rate (ORR), disease control rate (DCR), and incidence of severe adverse events (AEs); eligible Study designs (S) were clinical trials, excluding non-original research like basic research, retrospective studies, and case reports.
Inclusion criteria and exclusion criteria
Inclusion criteria: (I) a pathological assessment confirming a solid tumor; (II) treatment with MT plus ICI; (III) in every outcome, at least one of the following metrics was available or could be derived: ORR, DCR, PFS, OS, and serious AEs (SAEs) occurrences. Exclusion criteria: basic research, case reports, conference summaries, repeated publications of the same material by the same center and author, and non-original articles like meta-analyses and reviews, and analyses in which crucial data cannot be gathered.
Data extraction
The search strategy was collaboratively determined by all authors. Two researchers independently conducted the literature search, cross-referenced the retrieved literature, selected appropriate trials, and extracted data. During that time, if there was any disagreement, it was discussed by the team until a consensus was achieved. Collected data included the first author, publication year, country, clinical trial design description, sample size, evaluation criteria for efficacy and AEs, ORR, DCR, PFS, OS, and SAEs incidence.
Quality assessment
An independent evaluation of the literature’s quality was conducted by two researchers. The Methodological Index for Nonrandomised Studies (MINORS) scale, with a maximum of 16 points, was used to evaluate the methodological quality and bias risk of single-arm non-randomized trials. The evaluation included eight criteria: a clearly stated study aim, inclusion of consecutive patients, prospective data collection, and an endpoint suitable for the study; objective assessment of endpoints; follow-up duration suitable for the primary endpoint; less than 5% loss to follow-up; and pre-study determination of sample size, totaling 16 points. For randomized controlled trials (RCTs), the Jadad scale, totaling 4 points, assessed bias risk using criteria like randomization, allocation concealment, blinding, and withdrawal explanation. Two independent researchers scored the studies, resolving any disagreements through team discussions. The final quality and bias assessments were incorporated into the study’s qualitative synthesis and analysis.
Pooled data analysis
A qualitative synthesis of the eligible studies was presented in a table format, highlighting the research characteristics, clinical attributes, and reported efficacy and safety outcomes. This study analyzed heterogeneity across six included studies from both clinical and methodological perspectives. The findings revealed moderate to high heterogeneity, preventing quantitative meta-analysis and resulting in a qualitative synthesis instead. Clinically, heterogeneity was evident in tumor types (NSCLC, ovarian cancer, HER2-negative breast cancer), with NSCLC further divided into squamous/non-squamous and ovarian cancer into platinum-resistant/sensitive subgroups. Treatment lines varied from first to third-line, with some studies including patients from multiple lines. Intervention regimens featured four combinations, primarily vinorelbine + ICIs, along with triplet regimens like cyclophosphamide + ICIs + bevacizumab, and vinorelbine dosages ranged from 20–40 mg. Methodologically, the studies included 2 RCTs, 2 phase II single-arm trials, and 2 retrospective studies, with only two studies specifying the timing of ORR/DCR assessment.
Results
Eligible studies
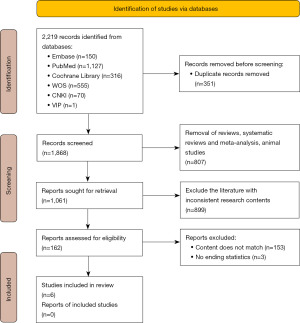
The literature screening followed the PRISMA statement’s recommendations. A total of 2,219 articles were retrieved from database searches, with contributions of 1,127 from PubMed, 316 from the Cochrane Library, 150 from Embase, 555 from Web of Science, and 70 from CNKI, 1 from VIP. We excluded 2,204 articles for reasons including duplicates, reviews, irrelevant topics, and in vitro study results. After a thorough review of the full texts, 15 articles were selected, and 9 were not included. Among the chosen articles, 2 could not be extracted, 3 were without outcome indicators, and 4 were non-MC plus ICI. The other 6 papers qualified (6,21-25). Figure 1 displays the flow chart, illustrating the literature search and study screening process.
Quality evaluation
The four trials were single-arm clinical studies, and their quality was assessed using the MINORS scale. The MINORS scores for the four studies ranged from 8 to 10 points (refer to Table 1 for detailed scores), suggesting a moderate overall quality with a low-to-moderate risk of bias. The Jadad scores for the two studies were 3 and 4 points, respectively, indicating a moderate-to-high overall quality with a low-to-moderate risk of bias. The Cochrane Risk of Bias tool 2.0 (RoB 2) was employed once more to assess the two RCTs. The results of this evaluation were in agreement with the Jadad scores, suggesting a low to moderate risk of bias. A comprehensive assessment of the six included studies revealed that biases were primarily associated with factors such as insufficient sample size, retrospective data collection, open-label design, and a slightly elevated loss to follow-up rate, resulting in an overall low-to-moderate risk of bias. Importantly, no studies with a high risk of bias were included.
Table1
| Author, year | Study type | Induction regimen | Cancer type | Line | Median age, years | Sample size | MINORS/Jadad score |
|---|---|---|---|---|---|---|---|
| Alain 2023 | II/non RCT | Atezolizumab (1,200 mg IV day 1, every 3 weeks) + oral vinorelbine (40 mg, 3 times by week) | NSCLC | Second-line | 64 | 71 | 9 |
| Chen 2024 | Retrospective | PD-1 + oral vinorelbine (40 mg, 3 times by week) for successive 3 weeks, and then discontinued for 1 week, with 28 days as a cycle | NSCLC | Second or later-line | 59 | 16 | 10 |
| Ruan 2024 | Retrospective | Oral vinorelbine (30 mg, 3 times by week) +sintilimab (200 mg IV day 1, every 3 weeks) | NSCLC | First line 22.64%; second line 41.51%; third or later-line 35.85% | 67 | 53 | 8 |
| Wan 2022 | II/RCT | Oral vinorelbine (20 mg, 3 times by week) +sintilimab (200 mg IV day 1, every 3 weeks) | NSCLC | Second or later-line | NA | 28 | 3 |
| Emese 2021 | II/non RCT | Intravenous pembrolizumab, 200 mg, and bevacizumab, 15 mg/kg, every 3 weeks and oral cyclophosphamide, 50 mg, once daily | Ovarian cancer | Second-line | 62.2 | 40 | 10 |
| Mo 2024 | II/RCT | Oral vinorelbine 40 mg, 3 times by week+ toripalimab 240 mg IV, every 21 d | Breast cancer | Second-line or first line | 47 | 7 | 4 |
| Oral cyclophosphamide 50 mg qd + oral capecitabine 500 mg tid + oral vinorelbine 40 mg 3 times by week+ toripalimab 240 mg IV, every 21 d | 50 | 26 | |||||
| Toripalimab 240 mg IV every 21 d+ bevacizumab 5 mg/kg IV every 21 d + oral vinorelbine 40 mg 3 times by week | 50 | 27 | |||||
| Oral vinorelbine 40 mg 3 times by week | 53 | 11 | |||||
| 50 mg/m2 IV every 21 d + oral vinorelbine 40 mg, 3 times by week + toripalimab 240 mg IV, every 21 d | 52 | 26 |
NA, not available; NSCLC, non-small cell lung cancer; PD-1, programmed cell death protein 1; RCT, randomized controlled trial.
Characteristics of the included studies
The study encompasses six clinical trials involving a total of 268 patient cases. Four of these trials employed a treatment regimen of ICI in conjunction with vinorelbine capsules. One trial utilized a regimen of ICI combined with cyclophosphamide tablets and bevacizumab. Another trial included three study groups with the following regimens: ICI plus vinorelbine capsules, ICI plus bevacizumab and vinorelbine capsules, and a combination of cyclophosphamide tablets, capecitabine tablets, vinorelbine capsules, and ICI. In all regimens, the chemotherapeutic agents were administered in a metronomic fashion. Detailed dosing regimens are presented in Table 1. Detailed ORR and DCR were documented in all six clinical trials. Five studies included detailed PFS data, three studies included detailed OS data, and five studies included detailed SAEs incidences. These specific outcomes are displayed in Table 2.
Table 2
| Author, year | mPFS (months) (95% CI) | mOS (months) (95% CI) | ORR (%) | DCR (%) | Evaluation criteria | Grade 3/4 AEs (%) | AE evaluation criteria |
|---|---|---|---|---|---|---|---|
| Alain 2023 | 2.2 (1.5–3.0) | 7.9 (4.8–11.4) | 4 months: 11 | 4 months: 32 | RECIST v1.1 | 5.10 | CTAE |
| Chen 2024 | 5.6 (4.8–6.4) | NA | 25 | 81.30 | RECIST v1.1 | 0.00 | CTCAE 5.0 |
| Ruan 2024 | 5.1 (4.2–6) | 8 (6–10) | 30.19 | 64.40 | RECIST v1.1 | 8.50 | CTCAE 5.0 |
| Wan 2022 | NA | NA | 4 cycles: 35.7 | 4 cycles: 75 | irRECIST | 0.00 | CTAE |
| Emese 2021 | 10.0 (6.5–17.4)† | NA | 47.50 | 95.00 | (irRECIST) or RECIST, v1.1 | 32.50 | CTAE |
| Mo 2024 | 1.2 (0.5–2.5) | 13.7 (5.1–30.3) | 0 | 26.4 | RECIST v1.1 | 12.00 | CTAE4.0 |
| 6.6 (4.0–10.4) | 42.6 (16.8–95.3) | 18.5 | 69.7 | 15.00 | |||
| 4.0 (2.4–5.9) | 47.7 (18.4–104.9) | 2.6 | 55.7 | 15.00 | |||
| 1.4 (0.7–2.4) | 19 (6.4–46.9) | 0 | 35.5 | 5.00 | |||
| 3.5 (2.2–5.3) | 23.1 (11.9–41.0) | 14.5 | 73.7 | 30.00 |
†, 90% CI. AE, adverse event; CI, confidence interval; DCR, disease control rate; mOS, median overall survival; mPFS, median progression-free survival; NA, not available; ORR, objective response rate.
The effectiveness and safety of MC + ICI in solid tumour
Vergnenegre et al. (23) conducted a multicenter, open-label phase II trial involving 71 NSCLC patients, with a median age of 64 years, 66.2% of whom were male, and most of whom were former or current smokers. Additionally, 90.2% of participants had an ECOG performance status of 0–1, and 83.1% had non-squamous NSCLC. After first-line treatment, they received atezolizumab (1,200 mg every 3 weeks) and oral vinorelbine (40 mg three times weekly). With a median follow-up of 8.1 months, the 4-month progression-free survival rate was 32%, with a median PFS of 2.2 months. The overall survival rates were 73.2% at 4 months and 24.3% at 24 months, with a median OS of 7.9 months. The 4-month ORR was 11%, and the DCR was 32%. Regarding safety, a total of 412 AEs were documented, among which 55 events (13.3%) were classified as grade 3–4 AEs. Of these, 21 events (5.1%) were attributed to the study treatment. The grade 3–4 AEs predominantly comprised diarrhea, vomiting, and anemia. Additionally, one grade 5 AE, resulting in death, was reported and attributed to pneumonia. The study conducted by Chen et al. (21) was a retrospective analysis evaluating the efficacy and safety of metronomic oral vinorelbine as a monotherapy compared to combination therapy regimens. The study included a total of 57 patients diagnosed with non-small cell lung cancer (NSCLC), with a median age of 59 years. The cohort comprised 36 males (63.2%) and 21 females (36.8%), with 68.4% of the patients having an Eastern Cooperative Oncology Group (ECOG) performance status of 2. Within the study, 12 patients received vinorelbine as monotherapy, 16 patients were treated with vinorelbine in combination with ICI, and 14 patients received vinorelbine in combination with anlotinib. Additionally, the group receiving vinorelbine in combination with epidermal growth factor receptor-tyrosine kinase inhibitors (EGFR-TKIs) included 15 patients. Across all groups, vinorelbine was administered orally at a dosage of 40 mg on three days per week (Monday, Wednesday, and Friday) for three consecutive weeks, followed by a 1-week rest period. The findings of the study demonstrated that the mPFS in the monotherapy cohort was 1.2 months, whereas the mPFS in the combination therapy cohorts was 4.6 months, a statistically significant improvement compared to the monotherapy group [hazard ratio (HR) =0.11, P<0.0001]. Subgroup analysis identified the vinorelbine plus immune checkpoint inhibitor regimen as exhibiting the most pronounced efficacy, with an mPFS of 5.6 months (95% CI: 4.8 to 6.4), an ORR of 25%, and a DCR of 81.3%. Regarding safety profiles, the monotherapy group reported three instances (25%) of grade 1–2 anemia, with no occurrences of grade 3–4 AEs. In contrast, the combination therapy group experienced 12 instances (40%) of grade 1–2 AEs and one instance (3.3%) of a grade 3 AE, with no grade 4 AEs reported. The predominant AEs in the combination therapy group included fatigue in two cases (6.7%), vomiting in two cases (6.7%), and anemia in eight cases (26.7%). Ruan’s (6) retrospective study involved 53 NSCLC patients with a median age of 67 years. The cohort comprised 67.92% males and 32.08% females, with 66.04% having a smoking history. ECOG performance status was 0 in 35.85%, 1 in 50.94%, and 2 in 13.21% of patients. Predominantly, 69.81% had squamous cell carcinoma. Treatment lines were first-line in 22.64%, second-line in 41.51%, and third-line or higher in 35.85%. EGFR wild type was observed in 47.17% of patients. PD-L1 expression was Tumor Proportion Score (TPS) ≥1% in 37.74%, TPS <1% in 18.87%, and unknown in 43.40%. Brain metastasis was present in 13.21% of patients. All patients received sintilimab (200 mg IV Q3W) and oral vinorelbine (30 mg tid, 3 days/week), completing at least 2 treatment cycles. The mPFS was 5.1 months, and the mOS was 8.0 months, 16 patients (30.19%) exhibited a partial response (PR), 17 patients (32.07%) maintained stable disease (SD), and 20 patients (37.74%) experienced progressive disease (PD). The overall DCR was calculated to be 64.4%, while the ORR was 30.19%. Among patients undergoing first-line, second-line, and third-line or higher treatments, the mPFS was 6.20, 5.25, and 3.70 months, respectively. Correspondingly, the mOS for these groups was 9.95, 8.10, and 6.00 months, respectively.
Regarding safety, a total of 199 AEs were identified, with the majority classified as grade 1–2 (182 out of 199), representing 91.5% of the total. The occurrence of grade ≥3 AEs was 8.5% (17 out of 199), predominantly comprising leukopenia (7.55%), nausea and vomiting (5.66%), anemia (5.66%), fatigue (3.77%), abnormal liver function (3.77%), abnormal renal function (1.89%), rash (1.89%), and pneumonia (1.89%). Treatment discontinuation occurred in only one patient due to a grade 3 adverse reaction (pneumonia), and there were no fatalities attributable to drug-related adverse reactions. The study conducted by Wan et al. (25) was an RCT that included 56 patients diagnosed with advanced NSCLC. Participants were randomly allocated into two groups: a monotherapy group and a combination therapy group, each comprising 28 patients. The monotherapy group received sintilimab at a dosage of 200 mg, administered intravenously over a period of 30 to 60 minutes, with a treatment cycle of 21 days. The combination therapy group was administered the same sintilimab regimen, supplemented with oral vinorelbine at a dosage of 20 mg, taken every other day, continuing until disease progression or intolerance, also adhering to a 21-day treatment cycle. The study’s findings revealed that after four treatment cycles, the ORR in the monotherapy group was 10.7%, with a DCR of 46.4%. In comparison, the combination therapy group demonstrated an ORR of 35.7% and a DCR of 75.0%. Both the ORR and DCR were significantly higher in the combination therapy group compared to the monotherapy group, with statistical significance (P<0.05). Adverse reactions in both cohorts were primarily classified as grade I–II and were generally well-tolerated by the patients. Notably, there were no instances of grade IV adverse reactions, and no patients discontinued treatment as a result of adverse reactions. Within the combination therapy group, the occurrences of nausea, vomiting, and leukopenia were significantly elevated compared to the monotherapy group, with these differences reaching statistical significance (P<0.05). The investigation conducted by Zsiros et al. (24) was designed as an open-label, single-arm, phase II clinical trial, enrolling a cohort of 40 individuals diagnosed with ovarian cancer. Within this cohort, 30 participants (75.0%) were identified as having platinum-resistant ovarian cancer, while the remaining 10 participants (25.0%) were classified as having platinum-sensitive ovarian cancer. The mean age of the participants was 62.2 years. The therapeutic protocol comprised the administration of intravenous pembrolizumab (200 mg) and bevacizumab (15 mg/kg) every 3 weeks, in conjunction with daily oral cyclophosphamide (50 mg), continued until the progression of the disease. The outcomes of the study demonstrated an ORR of 47.5% and a DCR of 95.0%, with 25.0% of the patients exhibiting durable responses persisting for more than 12 months. The mPFS was recorded at 10.0 months. Regarding safety, the most frequently observed grade 3–4 treatment-related AEs included hypertension, affecting 6 patients (15.0%), and lymphopenia, affecting 3 patients (7.5%). The study conducted by Mo et al. (22) was a phase II clinical trial utilizing a Bayesian adaptive randomization design, with dynamic allocation based on the DCR. A cohort of 97 patients with histologically or cytologically confirmed HER2-negative metastatic breast cancer was enrolled. The median age of the participants was 51 years. Among the patients, 63.9% had an ECOG performance status of 0, while 36.1% had a status of 1. Invasive ductal carcinoma was identified in 99.0% of the patients, whereas invasive lobular carcinoma was found in 1.0%. The histological grading of the tumors revealed that 2.1% were grade 1, 33.0% were grade 2, and 40.2% were grade 3. In terms of hormone receptor status, 50.5% of the patients were positive, and 49.5% were negative. HER2 expression was 0 in 40.2% of patients, 1 in 32.0%, and 2 in 27.8%. Regarding disease stage, 16.5% had primary stage IV, and 83.5% had recurrent or metastatic disease. Metastatic sites included lung (39.2%), liver (32.0%), bone (46.4%), brain (6.2%), chest wall (29.9%), and more than three sites (33.0%). Additionally, 42.3% had not received chemotherapy, while 57.7% had first-line chemotherapy. In this study, patients were randomly allocated into five distinct treatment groups: vinorelbine (NVB) monotherapy, NVB combined with toripalimab, a regimen of bevacizumab, NVB, and toripalimab (BEV group), a combination of cisplatin, NVB, and toripalimab (DDP group), and a regimen of metronomic cyclophosphamide, capecitabine, NVB, and toripalimab (VEX group). The findings demonstrated that the VEX and DDP groups exhibited the highest DCR, at 69.7% and 73.7%, respectively. The DCR in the BEV group was 55.7%, surpassing that of the NVB monotherapy and NVB plus toripalimab groups. The VEX group also achieved the longest mPFS of 6.6 months, followed by the BEV group at 4.0 months and the DDP group at 3.5 months. Notably, the BEV group recorded the longest mOS at 47.7 months, whereas the NVB monotherapy group had the shortest median OS at 19.0 months. The NVB plus toripalimab group had a median OS of 29.2 months. Regarding safety outcomes, the PD-1 + NVB cohort demonstrated a 12% incidence of grade 3 or higher adverse reactions. Within this group, immune-related pneumonia was observed in 5% of patients, immune hepatitis in 3%, hypothyroidism in 2%, and rash in 2%. The VEX cohort exhibited a 15% incidence of grade 3 or higher adverse reactions, with neutropenia occurring in 10% and elevated alanine aminotransferase levels in 5% of patients. Similarly, the BEV cohort reported a 15% incidence of grade 3 or higher adverse reactions, comprising hypertension in 5%, proteinuria in 5%, bleeding in 2%, thrombosis in 2%, and impaired wound healing in 1% of patients. The incidence of grade 3 or higher adverse reactions in the NVB monotherapy cohort was observed to be 5%, with neutropenia comprising the entirety of this percentage. In contrast, the DDP cohort exhibited a 30% incidence rate of grade 3 or higher adverse reactions, with neutropenia accounting for 20%, anemia for 10%, and nausea and vomiting for 5%. Significant differences in treatment response were noted among solid tumor subtypes. NSCLC had an ORR of 30.19% to 35.7% and a mPFS of 2.2 to 5.6 months. Breast cancer’s ORR ranged from 14.5% to 18.5%, with an mPFS of 3.5 to 6.6 months. Ovarian cancer showed the best outcomes, with an ORR of 47.5% and an mPFS of 10.0 months, attributed to a triplet regimen of MC, immune checkpoint inhibitors, and bevacizumab. The combination of anti-angiogenic agents with these treatments may enhance efficacy. Additionally, study design impacts outcomes; for instance, NSCLC’s ORR was 35.7% in an RCT (5) compared to 30.19% in a retrospective study (21), indicating that retrospective studies may underestimate efficacy due to selection bias. A strong link was found between therapy line and efficacy. NSCLC patients on first-line treatment had a mPFS of 6.20 months, much longer than the 3.70 months for those on third-line or later. This indicates that the MC + ICI combination is more effective when used earlier in solid tumor treatment.
Detailed information and data pertaining to these findings are presented in Tables 1,2.
Discussion
The six clinical studies incorporated into this research have demonstrated that patients with various tumor types and at different treatment stages maybe benefit from the combination of immunotherapy and MC. Furthermore, the efficacy of this combined approach appears to surpass that of MC alone. The findings indicate that the combination therapy is effective not only for patients undergoing initial treatment but also for those who have developed resistance or experienced relapse following multiple treatments, suggesting a potential synergistic interaction between the two modalities. The research mainly involved phase II/single-arm trials with 7 to 71 cases each, lacking statistical power and high-level phase III evidence. Thus, conclusions about the long-term efficacy of MC + ICI and their relevance to rare tumor types should be approached cautiously to prevent over-extrapolation. In recent years, there has been an increasing recognition that most tumors can modify their microenvironment to evade the immune system and reduce the effectiveness of standard treatment regimens (26). In the TME, cancer cells that engage in anaerobic metabolism exhibit a competitive advantage over cytotoxic lymphocytes in nutrient acquisition, thereby undermining the antitumor immune response. Furthermore, the administration of high doses of cytotoxic agents can lead to the depletion of immune cells, further compromising the functionality of the immune system (27). Many researches have demonstrated that immune checkpoint inhibitors can significantly improve therapeutic outcomes for cancer patients. The two most extensively studied targets in this context are the cytotoxic T-lymphocyte-associated antigen 4 (CTLA-4) and the PD-1 receptors, along with their corresponding soluble ligands, PD-L1 and PD-L2. By inhibiting these immune checkpoint molecules—namely CTLA-4, PD-1, and their ligands PD-L1 and PD-L2—immune checkpoint inhibitors facilitate the sustained activation of T cells. This mechanism prevents tumor cells from evading T cell activity through these checkpoints, thereby enhancing the immune system’s ability to recognize and eliminate tumors (28-32). MC represents a viable alternative to traditional chemotherapy regimens by employing lower doses of cytotoxic agents administered with increased frequency, as opposed to the conventional maximum tolerated dose (MTD) protocols. This approach does not primarily aim to directly target cancer cells; rather, it emphasizes the modulation of the supportive stromal environment. Stromal cells, including fibroblasts and pericytes, exhibit heightened susceptibility to chemotherapy and can be adversely affected by doses that remain non-toxic to malignant cells. By targeting the stroma, which serves as a source of angiogenic regulatory factors, MC seeks to inhibit tumor angiogenesis through the depletion of these angiogenic stimulatory factors (17-19). Basic research indicates that MC and immunotherapy can modulate the immune system through multiple pathways, exhibiting synergistic effects, with the specific regulatory mechanisms shown in Table 3 (26,33-36).
Table 3
| Variable | Metronomic chemotherapy | Immune checkpoint inhibitors |
|---|---|---|
| Enhance immune activation | Low-dose, high-frequency dosing minimizes damage to immune cells while enhancing the activation and functionality of CTLs and NK cells | By blocking immune checkpoint molecules, the activation of T cells is further maintained, thereby preventing tumor cells from inhibiting T cell function via these checkpoints |
| Mitigate nutritional competition | Reducing the number of tumor cells diminishes nutrient consumption, thereby providing immune cells with enhanced nutritional support and augmenting antitumor activity | Blocking PD-L1 on tumors can inhibit glycolysis, thereby providing cytotoxic lymphocytes with an additional competitive edge |
| Facilitate the infiltration of immune cells into tumors | Decreasing the quantity of immunosuppressive cells and the levels of immunosuppressive factors, while augmenting the expression of chemokines and adhesion molecules, facilitates the migration of immune cells to the tumor site | Augmenting the migration and infiltration of immune cells enables them to penetrate deeper into tumor tissues and directly target cancer cells |
CTL, cytotoxic T lymphocyte; NK, natural killer; PD-L1, programmed cell death ligand 1.
Preclinical investigations have demonstrated that the integration of MC with immune checkpoint inhibitors enhances immune cell activation, reduces nutritional competition, and facilitates immune cell infiltration into tumors, thereby improving the eradication of drug-resistant cancer cells. In vitro experiments involving NSCLC cell lines, irrespective of their cisplatin resistance or sensitivity, reveal that MC curtails the proliferation rate of NSCLC cells. The survival of a limited number of chemotherapy-sensitive cell lines following MC is sufficient to impede the growth rate of drug-resistant cells. Conversely, traditional MTD therapy suppresses the proliferation of drug-sensitive cells but fails to inhibit the growth of drug-resistant cells (37).
Our investigation identified four studies examining the application of immune checkpoint inhibitors in conjunction with MC for the treatment of NSCLC. Among these, three studies focused on populations receiving second-line or subsequent therapies, whereas the fourth study reported that 77% of its participants were in the second-line or higher treatment category. All four studies indicated that the combination of immunotherapy with metronomic vinorelbine chemotherapy exhibited a degree of efficacy. Nevertheless, the observed effectiveness varied among the studies, potentially due to differences in study design, variations in the populations included, and the intrinsic heterogeneity of the tumors. Studies by Vergnenegre et al. (23) and Ruan (6) show that patients with PD-L1 expression ≥1% benefit more from immunotherapy combined with MC. This aligns with findings from studies on immunotherapy with MTD chemotherapy. High PD-L1 expression may indicate stronger tumor immunogenicity, while MC can enhance the tumor immune microenvironment and decrease immunosuppressive cells, improving conditions for immune checkpoint inhibitors to activate T cells and eliminate tumor cells, resulting in synergistic effects.
Chen et al.’s study (13) demonstrated that the combination of metronomic vinorelbine and immunotherapy exhibited significant efficacy and good tolerability in patients with advanced NSCLC who tested negative for driver genes such as EGFR. Furthermore, in patients who were positive for driver genes and exhibited resistance to osimertinib, the regimen incorporating metronomic vinorelbine also demonstrated notable efficacy and tolerability, indicating its potential to overcome osimertinib resistance. Complementarily, the research conducted by Ruan (6) suggests that in patients with advanced NSCLC who are positive for driver genes, the combination of metronomic vinorelbine and immunotherapy is associated with a trend towards improved mPFS and mOS compared to those who are negative for driver genes. The underlying mechanism may involve MC’s capacity to inhibit angiogenesis in tumors harboring EGFR mutations and to disrupt the immunosuppressive characteristics of the TME. This suggests a novel therapeutic strategy for patients exhibiting resistance to targeted therapies.
Preclinical investigations conducted by Orlandi et al. have demonstrated that metronomic dosing of vinorelbine (VNR) significantly reduces the proliferation rate of two human NSCLC cell lines: one harboring a wild-type EGFR and the other possessing EGFR-TKI resistant mutations. Conversely, when vinorelbine was administered at the same dosage using the conventional MTD chemotherapy regimen, it exhibited efficacy solely in the EGFR wild-type NSCLC cell line (38). Furthermore, research by Wan et al. suggests that the efficacy of metronomic vinorelbine in combination with immunotherapy is comparable between adenocarcinoma and squamous cell carcinoma (25). Preclinical investigations have demonstrated that MC, when administered prior to immunotherapy in squamous cell lung cancer (SQCLC), exhibits enhanced synergistic efficacy with immunotherapy compared to conventional MTD chemotherapy. Low-dose chemotherapeutic agents are capable of inducing immunogenic cell death (ICD) in SQCLC cells through the activation of the PI3K/Akt/NF-κB signaling pathway, which subsequently enhances the exposure of neoantigens. This process leads to the release of high mobility group box 1 (HMGB1) from cells undergoing ICD, which in turn DCs to facilitate the uptake and presentation of neoantigens, thereby eliciting a specific T-cell response (39). Based on these findings, the combination of MC and immune checkpoint inhibitors demonstrates potential synergistic efficacy and promising clinical prospects in the treatment of NSCLC. Current research is constrained by limitations such as small sample sizes, ambiguous mechanisms, and the influence of heterogeneity. Further validation is required to identify optimal patient populations, potentially stratified by PD-L1/EGFR status, to optimize treatment protocols, including drug selection and dosing schedules, and to explore combination targeted therapies, such as those involving antiangiogenic agents. Future research directions should focus on conducting prospective stratified studies, elucidating underlying mechanisms, and extending applications to first-line and maintenance therapies. These efforts aim to provide less toxic and more effective strategies for the treatment of advanced NSCLC.
This study integrates clinical research findings that illustrate the efficacy of MC in conjunction with immunotherapy for the treatment of metastatic breast and ovarian cancers, particularly in cases where conventional chemotherapy has ceased to be effective. The investigation conducted by Zsiros et al. (24) demonstrated that the combination of metronomic cyclophosphamide and immunotherapy, further enhanced by bevacizumab, showed favorable tolerability and significant clinical efficacy in patients with recurrent ovarian cancer. This was evidenced by an ORR of 47.5% and a mPFS of 10.0 months. This multimodal therapeutic strategy may represent a novel treatment option for recurrent ovarian cancer. Ovarian cancer is characterized by high expression of vascular endothelial growth factor (VEGF), which is a key driver of tumor angiogenesis and local immune suppression. Bevacizumab specifically binds to and inhibits VEGF activity, thereby reducing tumor angiogenesis, improving the TME, enhancing T-cell infiltration, and decreasing the presence of immune-suppressive cells such as Tregs and myeloid-derived suppressor cells. This results in a synergistic triad of “immunotherapy-angiogenesis-chemotherapy”, which enhances therapeutic efficacy (40-42). The subgroup analysis of the study revealed that patients with platinum-sensitive ovarian cancer potentially experience greater benefits from the treatment. Conversely, patients exhibiting platinum resistance also demonstrated significant clinical responses. The ORR was notably higher in patients who were PD-L1 positive compared to those who were PD-L1 negative. However, the difference in PFS between these groups did not reach statistical significance (P=0.34). This indicates that while PD-L1 expression levels may influence treatment response to some extent, they are not a definitive determinant. Furthermore, patients who received three or fewer lines of chemotherapy showed significantly improved PFS and OS compared to those who underwent more than three lines of chemotherapy (P=0.03). This finding suggests that the early administration of the combination of immunotherapy, cyclophosphamide, and bevacizumab may result in superior therapeutic outcomes. The triplet regimen comprising MC (cyclophosphamide), immunotherapy, and bevacizumab has demonstrated significant efficacy in the treatment of recurrent ovarian cancer. This efficacy is attributed to the regimen’s synergistic antiangiogenic, immune-activating, and microenvironment-modulating effects, providing renewed hope particularly for patients exhibiting platinum resistance and those who have not responded to multiple prior treatments. The incorporation of bevacizumab not only augments antitumor activity but also potentially broadens the beneficiary population by enhancing immune infiltration and overcoming resistance. Future research endeavors should focus on conducting phase III RCTs to ascertain the optimal clinical scenarios for the application of this regimen across platinum-resistant, platinum-sensitive, and various treatment-line contexts. Additionally, it is imperative to integrate multi-omics analyses—encompassing factors such as PD-L1 expression, tumor mutational burden (TMB), immune microenvironment scores, and homologous recombination deficiency (HRD) status—to develop a composite biomarker model. This model would facilitate the prediction of therapeutic efficacy and enable precise identification of the patient population most likely to benefit from the treatment.
While ICIs have shown effectiveness across various cancer types, their use as monotherapy is limited in antitumor activity, particularly in immunologically cold tumors such as breast cancer (43). Currently, monotherapy chemotherapy remains the standard treatment for metastatic HER2-negative breast cancer; however, its efficacy is constrained and necessitates further improvement. Consequently, for patients with aggressive breast cancer, the combination of ICIs with chemotherapy or antiangiogenic agents may represent a clinically significant strategy. The integration of immunotherapy with cytotoxic agents has the potential to produce synergistic effects. Nonetheless, several challenges warrant consideration, including immune suppression induced by chemotherapy, the inherent toxicity of chemotherapeutic agents, patient tolerance, and the compounded toxicity resulting from the concurrent use of chemotherapy and immunotherapy. In the context of breast cancer, the efficacy of combining ICIs with various chemotherapy regimens exhibits notable variability. This variability may be attributed to the distinct characteristics of the TME engendered by different chemotherapeutic agents (44-46). The literature reviewed in this study indicates that a MC regimen, when combined with ICI treatment, exhibits substantial and durable efficacy in patients with metastatic breast cancer. This is particularly evident in the triple-negative breast cancer (TNBC) subgroup, where enhanced DCRs and progression-free survival have been observed. The study conducted by Mo et al. (22) demonstrated that the combination regimen of metronomic cyclophosphamide, capecitabine, and vinorelbine with immune checkpoint inhibitors resulted in superior DCRs and progression-free survival compared to the regimen of immune checkpoint inhibitors combined with vinorelbine alone in patients with metastatic breast cancer. The findings revealed significant differences in the immune cell subpopulations between responders and non-responders following treatment. Specifically, responders exhibited an increase in the proportion of certain immune cell subpopulations, whereas these subpopulations either remained unchanged or decreased in non-responders. This disparity suggests that MC may selectively activate and enhance the immune response, thereby improving therapeutic efficacy. The study utilized mass cytometry to investigate the effects of MC, revealing that this approach not only modified the proportions and functions of specific immune cell subpopulations, notably CD4+ T cells, monocytes, and NK cell subpopulations, but also induced a comprehensive reprogramming of the systemic immune response. This reprogramming was characterized by enhanced interactions among immune cells, increased activation status, and improved migration and positioning of immune cells, such as those expressing CCR4 and CCR7, thereby fostering a more favorable immune environment for immunotherapy. Research has demonstrated that cyclophosphamide exhibits immunosuppressive properties at high doses, whereas it exerts immunomodulatory effects when administered at metronomic doses. Specifically, it can selectively and significantly decrease the population of circulating Treg cells in cancer patients, thereby enhancing the antitumor response and cytotoxic capabilities of T lymphocytes and NK cells. Currently, MC with cyclophosphamide is employed either as a monotherapy or in combination with other anticancer agents, including antiangiogenic agents, immunomodulators, immune checkpoint inhibitors, vaccines, radiotherapy, and other conventional anticancer therapies, for the treatment of various cancer types as either first-line or subsequent-line interventions. Notably, over 75% of clinical studies have reported promising outcomes, particularly in breast, ovarian, and prostate cancers (47,48). MC using vinorelbine has demonstrated efficacy in the treatment of breast cancer. This therapeutic approach can delay the depletion of Treg cells while enhancing the activity of cytotoxic lymphocytes, thereby promoting adaptive immune responses. Consequently, its combination with ICIs may yield synergistic effects (49). Currently, several guidelines advocate for the use of MC in breast cancer management. However, there are no existing guidelines that recommend the combination of MC with immunotherapy (50,51).
The six studies incorporated in this research suggest that the combination of MC and immunotherapy is well-tolerated, with the majority of adverse reactions classified as grade I or II. In comparison to conventional maximum-tolerated-dose chemotherapy, MC is associated with relatively milder side effects and is more easily tolerated by patients. Furthermore, immune checkpoint inhibitors exhibit favorable tolerability, allowing even patients with slightly compromised physical conditions to adapt effectively to this treatment approach, thereby broadening the range of patients who can be treated. The advantageous safety profile suggests that patients may experience fewer adverse reactions during treatment, enabling them to better maintain their daily activities and work, thus significantly enhancing their quality of life. Ultimately, as research in this area continues to advance and optimize treatment protocols and manage adverse reactions, it is expected to improve therapeutic efficacy, reduce treatment-related risks, and extend the benefits of this combined therapeutic strategy to a larger patient population (52).
While the combination of ICIs and MC is generally well-tolerated, severe AEs are a concern, with grade ≥3 AEs occurring in 3.3% to 32.5% of cases across six studies. Regimens with bevacizumab or cisplatin had over 25% grade ≥3 AEs rates, while those with vinorelbine and an ICI had rates of 3.3–8.5%. Severe AEs mainly affected the hematologic system (leukopenia 7.55%, anemia 5.66%), cardiovascular system (hypertension 15.0%), and included immune-related pneumonia (1.89–5%). All severe AEs were manageable, with only one treatment discontinuation due to pneumonia and no treatment-related deaths. The safety profile was notably better than traditional high-dose chemotherapy, highlighting the need for stratified management based on regimen characteristics in clinical practice. Patients with cardiovascular diseases should cautiously use bevacizumab regimens. Regular monitoring of blood counts, blood pressure, and other indicators is crucial to promptly address severe side effects and optimize the treatment’s risk-benefit ratio.
The six studies included in this research generally showed a low-to-moderate risk of bias, though some bias factors might slightly affect the accuracy of efficacy results. Small sample sizes, especially in subgroup analyses like PD-L1 status or therapy lines, may lead to inadequate statistical power, potentially skewing efficacy outcomes. For instance, the Ruan’s study (6) reported a higher ORR in patients with PD-L1 TPS ≥1% compared to those with TPS <1%, but with only 20 PD-L1 positive patients, this finding might be due to random error and needs confirmation in larger studies. Second, bias from retrospective data collection in the two non-RCTs may lead to incomplete clinical information, impacting efficacy indicators. For example, the Chen et al. 2024 study (21) did not fully report EGFR mutation subtype distribution, despite evidence of differing chemotherapy responses between EGFR 19del and L858R patients. This omission could result in inaccurate conclusions about the benefits for patients with driver gene mutations. Detection bias from the open-label design (no blinding in the two RCTs) might affect the assessment of efficacy endpoints. While objective criteria like RECIST v1.1/irRECIST were used for ORR and DCR, subjective endpoints (e.g., quality of life) weren’t analyzed, and AEs (e.g., fatigue, nausea) might be slightly overestimated without blinding. However, this bias minimally impacts core efficacy indicators (tumor size changes, survival time) as they depend less on subjective judgment.
Limitations
The study’s bias does not undermine the main conclusion that MC with immunotherapy benefits patients with solid tumors unresponsive to conventional treatments, but it does restrict the generalizability of the findings.
Nevertheless, due to the limited number of extant studies, future research should prioritize large-scale, multicenter clinical trials to validate efficacy and safety, explore immune mechanisms in greater depth, develop predictive biomarkers for personalized treatment, optimize combination therapy regimens, and balance therapeutic efficacy with safety. The ultimate objective is to provide improved treatment options for breast cancer patients. The investigation of MC in conjunction with immunotherapy for the treatment of solid tumors is progressively gaining traction, although the majority of research remains in its early stages. For example, Zhou et al. (53) conducted a phase Ib, open-label, single-center, multi-cohort study to assess the safety and efficacy of camrelizumab combined with metronomic capecitabine in patients with refractory solid tumors. Additionally, emerging evidence suggests that this therapeutic combination may be promising for colorectal cancer; however, current studies are limited to preclinical models, with clinical trials still in progress (40). Numerous ongoing studies, including NCT03879512, NCT03585465, NCT04535713, NCT04984733, NCT03425292, NCT04331626, and NCT03349450, are further exploring this treatment strategy. As research advances, the integration of MC and immunotherapy has the potential to provide novel therapeutic options and renewed hope for a wider spectrum of patients with solid tumors.
Conclusions
The integration of MC with immune checkpoint inhibitors presents significant potential in the realm of cancer treatment. This approach not only augments therapeutic efficacy and expands the range of applicable cases but also enhances patients’ quality of life and accelerates the progress of precision medicine. As research further investigates the underlying mechanisms of this combined therapeutic strategy, it is expected that a variety of biomarkers predictive of treatment outcomes will be identified. These biomarkers will empower clinicians to accurately determine, before initiating treatment, the patient cohorts most likely to benefit from the combination therapy, thereby advancing the realization of precision medicine and providing cancer patients with increased hope and opportunities.
Acknowledgments
None.
Footnote
Reporting Checklist: The authors have completed the PRISMA reporting checklist. Available at https://tcr.amegroups.com/article/view/10.21037/tcr-2025-1-2912/rc
Peer Review File: Available at https://tcr.amegroups.com/article/view/10.21037/tcr-2025-1-2912/prf
Funding: None.
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://tcr.amegroups.com/article/view/10.21037/tcr-2025-1-2912/coif). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Filho AM, Laversanne M, Ferlay J, et al. The GLOBOCAN 2022 cancer estimates: Data sources, methods, and a snapshot of the cancer burden worldwide. Int J Cancer 2025;156:1336-46. [Crossref] [PubMed]
- Siegel RL, Giaquinto AN, Jemal A. Cancer statistics, 2024. CA Cancer J Clin 2024;74:12-49. [Crossref] [PubMed]
- Wang R, Guo T, Wang Q, et al. The efficacy and safety of induction chemotherapy combined with sintilimab followed by concurrent chemoradiotherapy plus sintilimab sequencing maintaining with sintilimab for patients with unresectable locally advanced esophageal squamous cell carcinoma. BMC Cancer 2025;25:97. [Crossref] [PubMed]
- Sim ES, Nguyen HCB, Hanna GJ, et al. Current Progress and Future Directions of Immunotherapy in Head and Neck Squamous Cell Carcinoma: A Narrative Review. JAMA Otolaryngol Head Neck Surg 2025;151:521-8. [Crossref] [PubMed]
- Zsiros E, Frederick PJ, Akers SN, et al. A phase II trial of pembrolizumab in combination with bevacizumab and oral metronomic cyclophosphamide for recurrent epithelial ovarian, fallopian tube or primary peritoneal cancer. Gynecologic Oncology 2019;154:23.
- Ruan Y. Effectiveness and safety of Sintilimab combined with Metronomic Oral Vinorelbine in the treatment of advanced non-small cell lung cancer (NSCLC). Zhejiang Chinese Medical University 2024;
- Maharjan R, Choi JU, Kweon S, et al. A novel oral metronomic chemotherapy provokes tumor specific immunity resulting in colon cancer eradication in combination with anti-PD-1 therapy. Biomaterials 2022;281:121334. [Crossref] [PubMed]
- Rajczykowski M, Olbryt M, Galwas K, et al. Sustained Immunotherapy Response in Metastatic Brain Melanoma Through 2 Pregnancies. Am J Case Rep 2025;26:e945533. [Crossref] [PubMed]
- Park J, Joung JG, Lim MC, et al. Neoadjuvant Chemotherapy with Dual Immune Checkpoint Inhibitors for Advanced-Stage Ovarian Cancer: Final Analysis of TRU-D Phase II Nonrandomized Clinical Trial. Clin Cancer Res 2025;31:1865-76. [Crossref] [PubMed]
- Othman T, Frankel P, Allen P, et al. Atezolizumab combined with immunogenic salvage chemoimmunotherapy in patients with transformed diffuse large B-cell lymphoma. Haematologica 2025;110:142-52. [Crossref] [PubMed]
- André N, Kerbel RS. Metronomic chemotherapy and drug repurposing: Two sides of an oncology coin to reduce host and financial toxicity. Cancer Lett 2025;613:217437. [Crossref] [PubMed]
- Chen L, Cao X, Li J, et al. Efficacy and safety of metronomic chemotherapy in maintenance therapy for metastatic colorectal cancer: A systematic review of randomized controlled trials. Medicine (Baltimore) 2022;101:e31659. [Crossref] [PubMed]
- Chen X, He Y, Fan T, et al. Efficacy and Safety of Low-Dose Oral Etoposide Combined With Capecitabine for Patients With Postoperative Metastatic Breast Cancer Resistant to Anthracycline/Taxanes. Thorac Cancer 2025;16:e70003. [Crossref] [PubMed]
- Sugihara T, Tsujimoto H, Itazaki Y, et al. Metronomic Photodynamic Therapy With Immune Checkpoint Inhibitors Does Not Affect Nontarget Lesions. J Surg Res 2025;308:9-18. [Crossref] [PubMed]
- Li G, Jiang Y, Tong H, et al. Sciatic nerve stimulation enhances NK cell cytotoxicity through dopamine signaling and synergizes immunotherapy in triple-negative breast cancer. Drug Resist Updat 2025;79:101212. [Crossref] [PubMed]
- Cai XJ, Fei WD, Xu YY, et al. Combination of metronomic administration and target delivery strategies to improve the anti-angiogenic and anti-tumor effects of triptolide. Drug Deliv Transl Res 2020;10:93-107. [Crossref] [PubMed]
- You R, Wang B, Chen P, et al. Metformin sensitizes AML cells to chemotherapy through blocking mitochondrial transfer from stromal cells to AML cells. Cancer Lett 2022;532:215582. [Crossref] [PubMed]
- Su F, Wang X, Pearson T, et al. Ablation of Stromal Cells with a Targeted Proapoptotic Peptide Suppresses Cancer Chemotherapy Resistance and Metastasis. Mol Ther Oncolytics 2020;18:579-86. [Crossref] [PubMed]
- Matula Z, Mikala G, Lukácsi S, et al. Stromal Cells Serve Drug Resistance for Multiple Myeloma via Mitochondrial Transfer: A Study on Primary Myeloma and Stromal Cells. Cancers (Basel) 2021;13:3461. [Crossref] [PubMed]
- Yamazaki Y, Tezuka Y, Ono Y, et al. Updates on WHO 5th edition classification, molecular characteristics and tumor microenvironment of adrenocortical carcinomas. Endocr J 2025;72:243-57.
- Chen S, He Z, Li M, et al. Efficacy and safety of metronomic oral vinorelbine and its combination therapy as second- and later-line regimens for advanced non-small-cell lung cancer: a retrospective analysis. Clin Transl Oncol 2024;26:3202-10. [Crossref] [PubMed]
- Mo H, Yu Y, Sun X, et al. Metronomic chemotherapy plus anti-PD-1 in metastatic breast cancer: a Bayesian adaptive randomized phase 2 trial. Nat Med 2024;30:2528-39. [Crossref] [PubMed]
- Vergnenegre A, Monnet I, Ricordel C, et al. Safety and efficacy of second-line metronomic oral vinorelbine-atezolizumab combination in stage IV non-small-cell lung cancer: An open-label phase II trial (VinMetAtezo). Lung Cancer 2023;178:191-7. [Crossref] [PubMed]
- Zsiros E, Lynam S, Attwood KM, et al. Efficacy and Safety of Pembrolizumab in Combination With Bevacizumab and Oral Metronomic Cyclophosphamide in the Treatment of Recurrent Ovarian Cancer: A Phase 2 Nonrandomized Clinical Trial. JAMA Oncol 2021;7:78-85. [Crossref] [PubMed]
- Wan X, Fang M, Zhou Q, et al. Observation on the efficacy of PD-1 inhibitor combined with metronomic chemotherapy in the treatment of advanced non-small cell lung cancer. Proceeding of Clinical Medicine 2022;31:341-4.
- Chang CH, Qiu J, O'Sullivan D, et al. Metabolic Competition in the Tumor Microenvironment Is a Driver of Cancer Progression. Cell 2015;162:1229-41. [Crossref] [PubMed]
- Kareva I, Berezovskaya F. Cancer immunoediting: A process driven by metabolic competition as a predator-prey-shared resource type model. J Theor Biol 2015;380:463-72. [Crossref] [PubMed]
- Janjigian YY, Ajani JA, Moehler M, et al. First-Line Nivolumab Plus Chemotherapy for Advanced Gastric, Gastroesophageal Junction, and Esophageal Adenocarcinoma: 3-Year Follow-Up of the Phase III CheckMate 649 Trial. J Clin Oncol 2024;42:2012-20. [Crossref] [PubMed]
- Verschoor YL, van de Haar J, van den Berg JG, et al. Neoadjuvant atezolizumab plus chemotherapy in gastric and gastroesophageal junction adenocarcinoma: the phase 2 PANDA trial. Nat Med 2024;30:519-30. [Crossref] [PubMed]
- Oaknin A, Gladieff L, Martínez-García J, et al. Atezolizumab plus bevacizumab and chemotherapy for metastatic, persistent, or recurrent cervical cancer (BEATcc): a randomised, open-label, phase 3 trial. Lancet 2024;403:31-43. [Crossref] [PubMed]
- Ready NE, Ott PA, Hellmann MD, et al. Nivolumab Monotherapy and Nivolumab Plus Ipilimumab in Recurrent Small Cell Lung Cancer: Results From the CheckMate 032 Randomized Cohort. J Thorac Oncol 2020;15:426-35. [Crossref] [PubMed]
- Wolchok JD, Chiarion-Sileni V, Rutkowski P, et al. Final, 10-Year Outcomes with Nivolumab plus Ipilimumab in Advanced Melanoma. N Engl J Med 2025;392:11-22. [Crossref] [PubMed]
- Pepe FF, Cazzaniga ME, Baroni S, et al. Immunomodulatory effects of metronomic vinorelbine (mVRL), with or without metronomic capecitabine (mCAPE), in hormone receptor positive (HR+)/HER2-negative metastatic breast cancer (MBC) patients: final results of the exploratory phase 2 Victor-5 study. BMC Cancer 2022;22:956. [Crossref] [PubMed]
- Wu J, Waxman DJ. Metronomic cyclophosphamide eradicates large implanted GL261 gliomas by activating antitumor Cd8(+) T-cell responses and immune memory. Oncoimmunology 2015;4:e1005521. [Crossref] [PubMed]
- Wu J, Waxman DJ. Metronomic cyclophosphamide schedule-dependence of innate immune cell recruitment and tumor regression in an implanted glioma model. Cancer Lett 2014;353:272-80. [Crossref] [PubMed]
- Kareva I. A Combination of Immune Checkpoint Inhibition with Metronomic Chemotherapy as a Way of Targeting Therapy-Resistant Cancer Cells. Int J Mol Sci 2017;18:2134. [Crossref] [PubMed]
- Bondarenko M, Le Grand M, Shaked Y, et al. Metronomic Chemotherapy Modulates Clonal Interactions to Prevent Drug Resistance in Non-Small Cell Lung Cancer. Cancers (Basel) 2021;13:2239. [Crossref] [PubMed]
- Orlandi P, Di Desidero T, Salvia G, et al. Metronomic vinorelbine is directly active on Non Small Cell Lung Cancer cells and sensitizes the EGFR(L858R/T790M) cells to reversible EGFR tyrosine kinase inhibitors. Biochem Pharmacol 2018;152:327-37. [Crossref] [PubMed]
- He XR, Du Y, Jin YS, et al. Upfront metronomic chemotherapy synergizes with immunotherapy to optimize chemoimmunotherapy in squamous cell lung carcinoma. Cancer research 2020;80:Abstract nr 2207.
- Hong JH, Woo IS. Metronomic chemotherapy as a potential partner of immune checkpoint inhibitors for metastatic colorectal cancer treatment. Cancer Lett 2023;565:216236. [Crossref] [PubMed]
- Joshi S, Durden DL. Combinatorial Approach to Improve Cancer Immunotherapy: Rational Drug Design Strategy to Simultaneously Hit Multiple Targets to Kill Tumor Cells and to Activate the Immune System. J Oncol 2019;2019:5245034. [Crossref] [PubMed]
- Xu Q, Shao D. Leveraging the synergy between anti-angiogenic therapy and immune checkpoint inhibitors to treat digestive system cancers. Front Immunol 2024;15:1487610. [Crossref] [PubMed]
- Adams S, Loi S, Toppmeyer D, et al. Pembrolizumab monotherapy for previously untreated, PD-L1-positive, metastatic triple-negative breast cancer: cohort B of the phase II KEYNOTE-086 study. Ann Oncol 2019;30:405-11. [Crossref] [PubMed]
- Cortes J, Rugo HS, Cescon DW, et al. Pembrolizumab plus Chemotherapy in Advanced Triple-Negative Breast Cancer. N Engl J Med 2022;387:217-26. [Crossref] [PubMed]
- Miles D, Gligorov J, André F, et al. Primary results from IMpassion131, a double-blind, placebo-controlled, randomised phase III trial of first-line paclitaxel with or without atezolizumab for unresectable locally advanced/metastatic triple-negative breast cancer. Ann Oncol 2021;32:994-1004. [Crossref] [PubMed]
- Dent R, André F, Gonçalves A, et al. IMpassion132 double-blind randomised phase III trial of chemotherapy with or without atezolizumab for early relapsing unresectable locally advanced or metastatic triple-negative breast cancer. Ann Oncol 2024;35:630-42. [Crossref] [PubMed]
- Delahousse J, Molina L, Paci A. Cyclophosphamide and analogues; a matter of dose and schedule for dual anticancer activities Cancer Lett 2024;598:217119. [Crossref] [PubMed]
- Røssevold AH, Andresen NK, Bjerre CA, et al. Atezolizumab plus anthracycline-based chemotherapy in metastatic triple-negative breast cancer: the randomized, double-blind phase 2b ALICE trial. Nat Med 2022;28:2573-83. [Crossref] [PubMed]
- Freyer G, Martinez-Jañez N, Kukielka-Budny B, et al. Single-agent metronomic versus weekly oral vinorelbine as first-line chemotherapy in patients with HR-positive/HER2-negative advanced breast cancer: The randomized Tempo Breast study. Breast 2024;74:103681. [Crossref] [PubMed]
- Cardoso F, Paluch-Shimon S, Senkus E, et al. 5th ESO-ESMO international consensus guidelines for advanced breast cancer (ABC 5). Ann Oncol 2020;31:1623-49. [Crossref] [PubMed]
- Breast Cancer Expert Committee of National Cancer Quality Control Center. Cancer Drug Clinical Research Committee of China Anti-Cancer Association. Guidelines for clinical diagnosis and treatment of advanced breast cancer in China (2020 Edition). Zhonghua Zhong Liu Za Zhi 2020;42:781-97.
- Yoon JS, Zhao F, Masood H, et al. Comparative study of management strategies for immune checkpoint inhibitor-induced inflammatory arthritis: rheumatologists versus oncologists. Intern Med J 2025;55:426-34. [Crossref] [PubMed]
- Zhou C, Shangguan C, Shi M, et al. Camrelizumab and metronomic capecitabine for patients with treatment-refractory solid tumors (McCREST trial). Future Oncol 2022;18:2495-503. [Crossref] [PubMed]